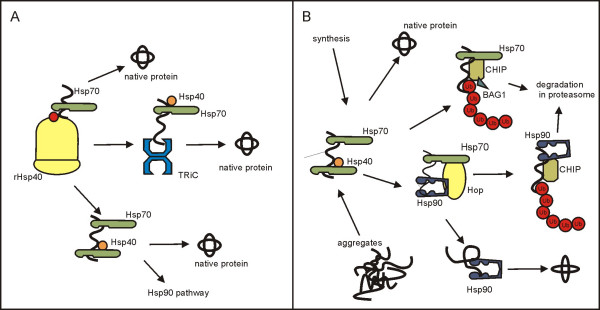
Figure 2.
Chaperone-assisted folding of proteins in eukaryotes [34].(A) Early phase of folding of newly synthesized proteins. Hsp40 molecules attached to ribosomes (rHsp40) activate cytosolic Hsp70 molecules and direct them to elongating polypeptides. This co-translational chaperoning may be sufficient for the correct folding of many proteins. Folding of some others is assisted by Hsp70 also post-translationally. This can require collaboration of Hsp70 (or prefoldin, not shown) with TRiC. Finally, Hsp70 can direct some proteins to Hsp90, which initiates the late phase of folding. (B) Late phase of protein folding and protein quality control in eukaryotes. A protein held by Hsp70 can be released and free from further assistance of the chaperones providing that it manages to adopt a proper conformation. In case of excessively unstable proteins, Hsp70 can recruit the co-chaperones CHIP and BAG1 and this eventually leads to protein degradation in the proteasome. Some special proteins are transferred from Hsp70 to Hsp90 with the help of the co-chaperone HOP. Successfully transferred protein is held by Hsp90 which enables its final modification; alternatively, the protein is directed to proteolysis. The binding by Hsp70 is typical both for newly synthesized and secondarily misfolded polypeptides. If the latter happens, the binding of Hsp70 to polypeptides is governed by Hsp40 chaperones other than those attached to ribosomes. This is one way to differentiate the specificity and intensity of the eukaryotic chaperone activity between normal metabolism (protein synthesis) and stress reaction (protein reactivation).