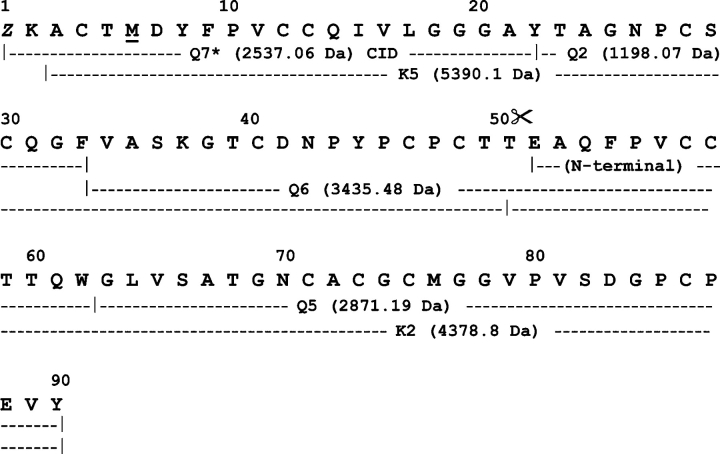
Figure 4.
Amino acid sequence of HCA. The primary structures of HCA were determined by combination of Edman degradation of sets of overlapping peptides obtained by proteolysis of the reduced and carbamidomethylated full-length lectin and its reversed-phase HPLC fragments (isolated as in Fig. 3 ▶) with chymotrypsin (Q-) and endoproteinase Lys-C (K-), and by CID MS/MS analysis of the N-terminal blocked peptide Q7. Methionine residue at position 6 was oxidized in Q7. Proteolysis at the Thr50–Glu51 peptide bond, which generates the N-terminal sequence determined in the native two-chain HCA lectin and in its 4390-Da C-terminal fragment (identical to peptide K2), is indicated by scissors. (Z) Pyroglutamic acid.