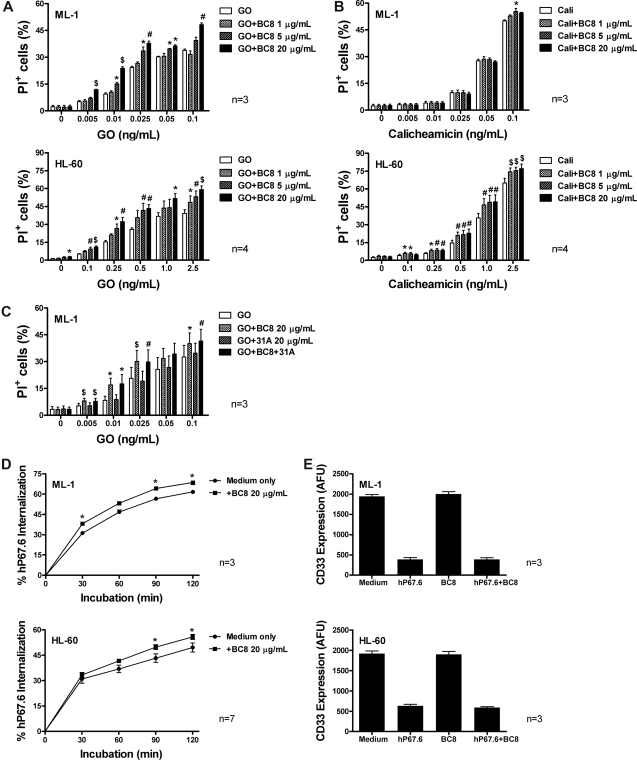
Figure 1.
Effect of BC8 on GO- and calicheamicin-γ1–induced cytotoxicity as well as CD33 internalization and modulation in human AML cell lines in vitro. (A,B) Drug-induced cytotoxicity. ML-1 and HL-60 cells (top and bottom panels, respectively) were incubated with various concentrations of (A) GO or (B) calicheamicin-γ1 for 3 days in the presence or absence of increasing concentrations of BC8. Cytotoxicity was assessed using PI staining and expressed as the percentage of PI+ cells. (C) GO-induced cytotoxicity in presence of BC8 and 31A. ML-1 cells were incubated with various concentrations of GO for 3 days in the presence or absence of BC8 (20 μg/mL) and/or 31A (20 μg/mL). (D) CD33 endocytosis. ML-1 and HL-60 cells (top and bottom panels, respectively) were incubated for 30 minutes with medium containing 2.5 μg/mL unconjugated, unlabeled hP67.6 in ice water to prevent internalization during the staining procedure. Cells were then washed in ice-cold PBS to remove unbound antibody, resuspended in antibody-free medium, and incubated at 37°C (in 5% CO2 and air) for various periods of time. Subsequently, cells were chilled and incubated with biotin-conjugated mouse anti–human IgG4 monoclonal antibody (5 μg/mL), followed by incubation with streptavidin-PE conjugate (5 μg/mL) to detect remaining hP67.6 on the cell surface. One sample that was kept in ice water was used to determine the starting level of antibody bound to the cell. (E) CD33 modulation. ML-1 and HL-60 cells (top and bottom panels, respectively) were incubated overnight in the presence or absence of hP67.6 and/or BC8. Cell surface CD33 was then measured by subsequent staining with hP67.6, a biotin-conjugated mouse anti–human IgG4 antibody, and a streptavidin-PE conjugate. Results are expressed as arbitrary fluorescence units (AFU) and shown as mean plus or minus SEM from 3 to 7 independent experiments. *P < .05; #P < .01; $P < .001.