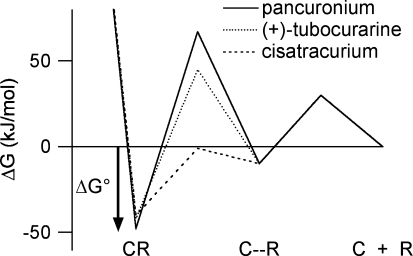
Figure 7.
A free energy diagram describing the binding of competitive antagonists to the nAChR in an induced-fit model The energy well on the right corresponds to a weakly bound antagonist–receptor encounter complex (C–R). It is reached by overcoming a diffusional barrier that is assumed to be the same for all antagonists (30 kJ mol−1). Similarly, the activation energy for dissociation away from this state is assumed to be antagonist independent (40 kJ mol−1). The barrier between the encounter complex and the equilibrium bound state reflects a conformational change (induced fit) in the receptor protein and depends on the antagonist. The activation energies for this barrier are calculated as the difference between the measured activation energies (Tables 3 and 4) and the energies assumed for the diffusional barrier (Shoup & Szabo, 1982).