Abstract
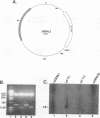
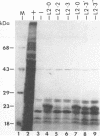
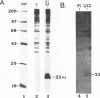
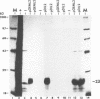
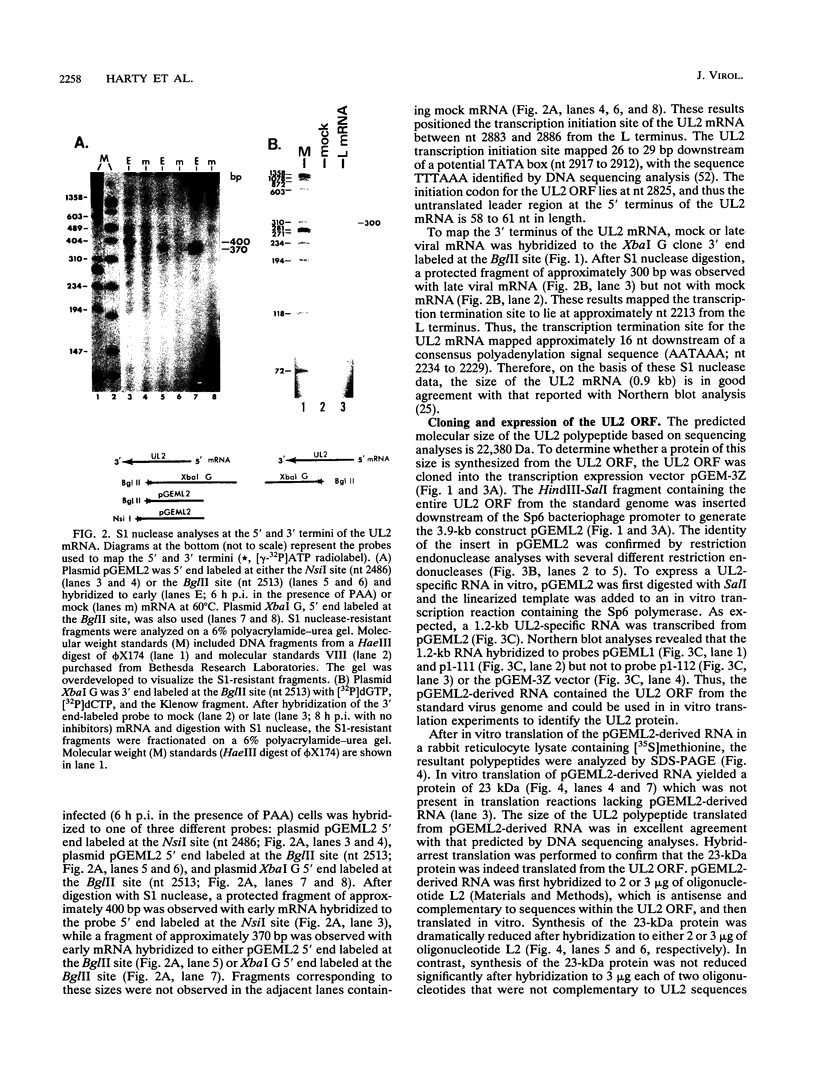
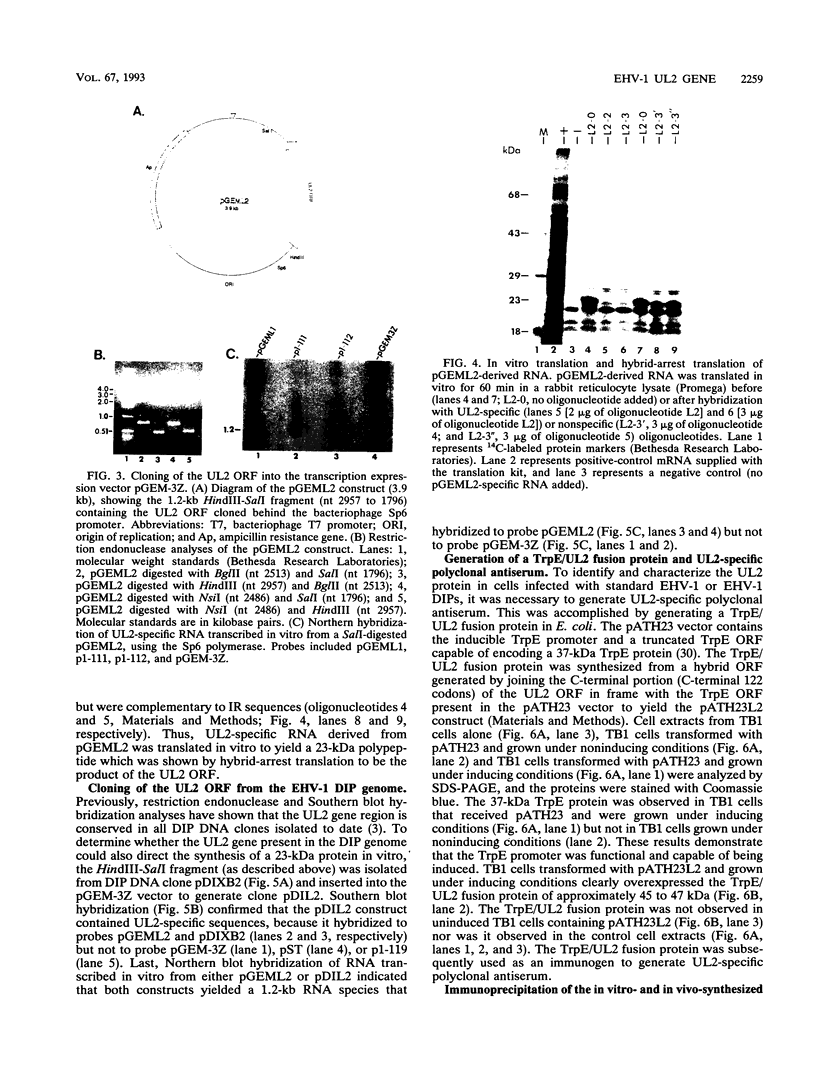
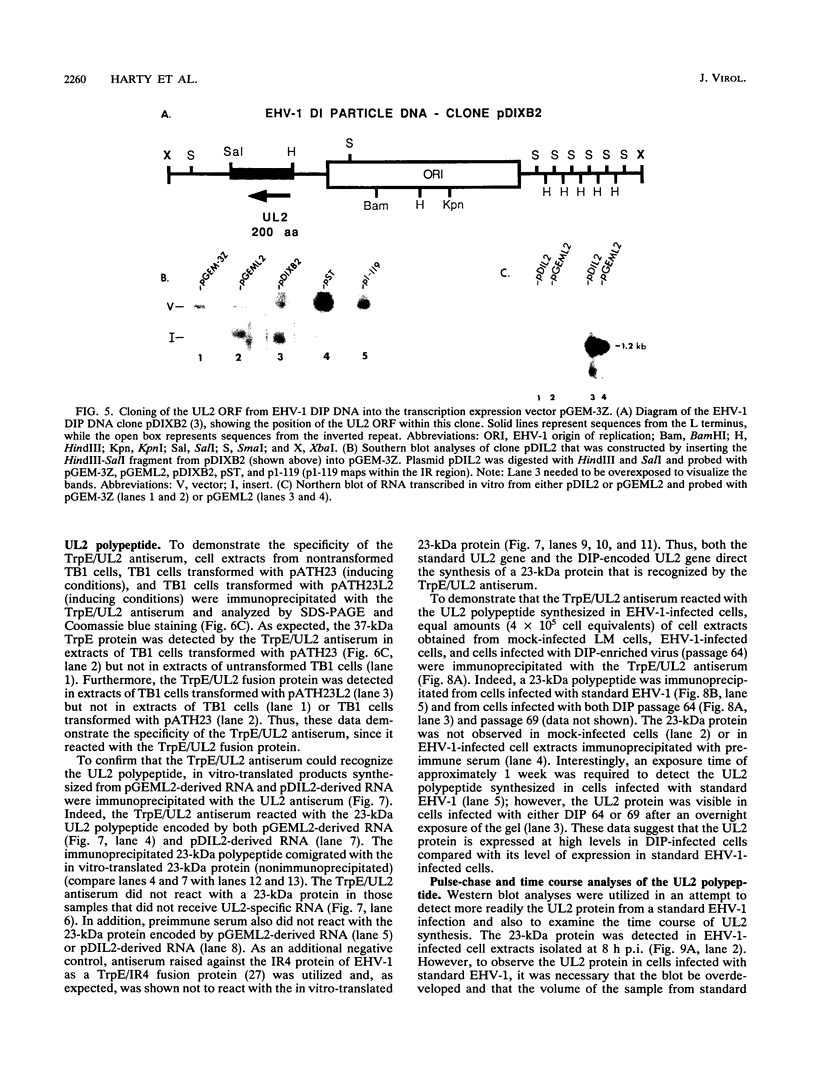
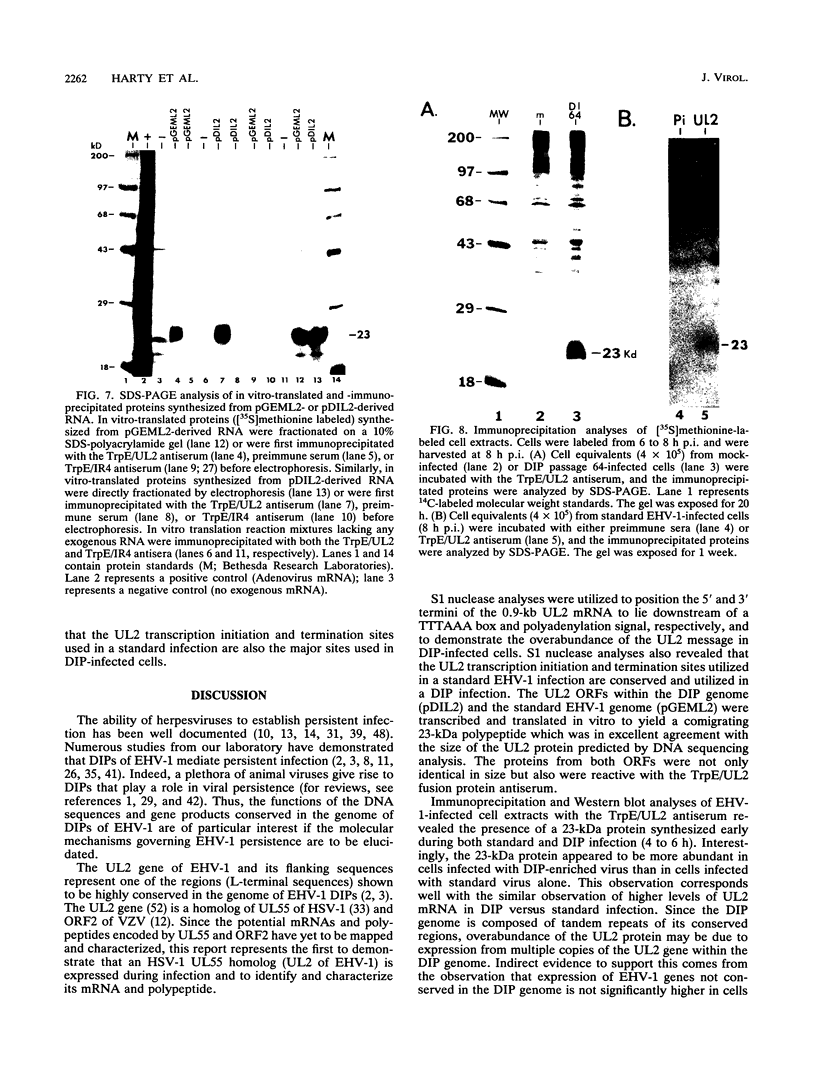
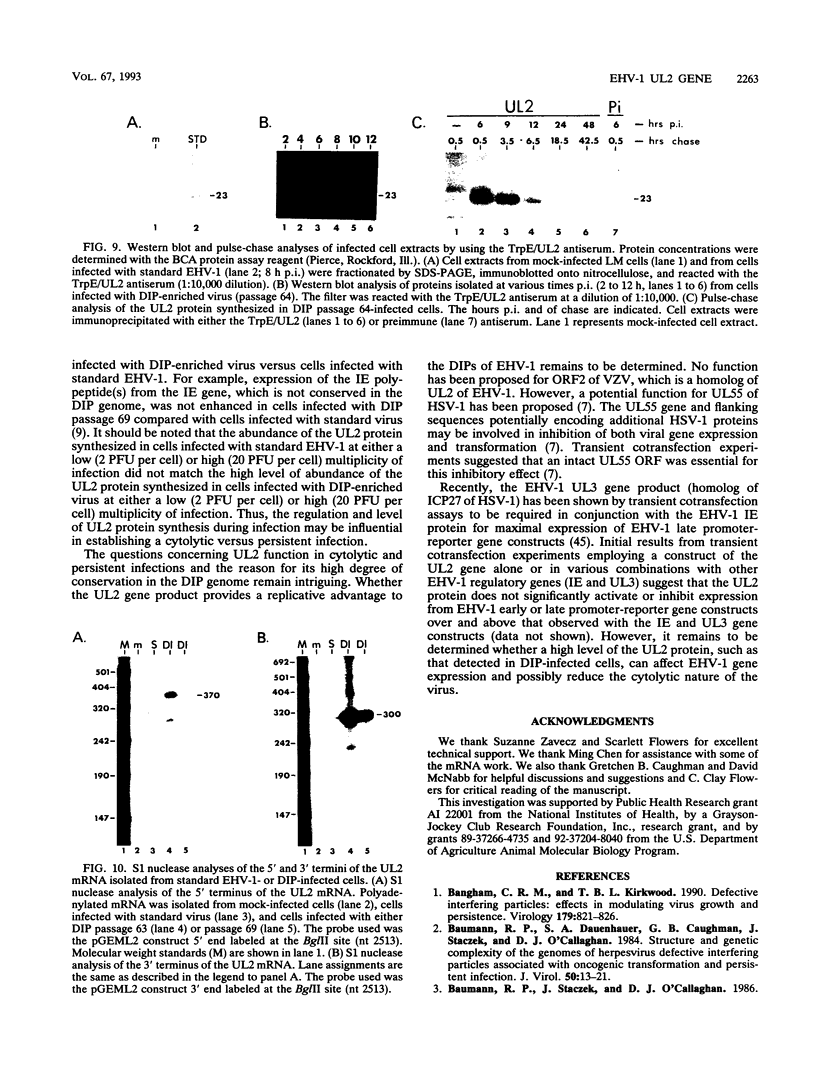
Defective interfering particles (DIPs) of equine herpesvirus 1 (EHV-1; Kentucky A strain) mediate persistent infection. DNA sequences at the L terminus, which contain the UL2 gene (homolog of UL55 of herpes simplex virus type 1 and open reading frame 3 of varicella-zoster virus) of standard EHV-1, have been shown to be highly conserved in all clones of the EHV-1 DIP genome. The UL2 mRNA was characterized by S1 nuclease analyses, which mapped the 5' and 3' termini of the 0.9-kb early UL2 mRNA to approximately 26 and 16 nucleotides downstream of a TTTAAA box and polyadenylation signal, respectively. The UL2 open reading frame, present within both the EHV-1 standard and DIP genomes, was inserted into the transcription expression vector pGEM-3Z to yield constructs pGEML2 and pDIL2, respectively. After in vitro transcription and translation, both constructs yielded a comigrating 23-kDa protein, as determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Polyclonal antiserum was raised against the UL2 protein by injecting rabbits with a TrpE/UL2 fusion protein expressed from plasmid pATH23L2 in Escherichia coli. The UL2-specific antiserum reacted in Western immunoblot and immunoprecipitation analyses with a 23-kDa polypeptide synthesized in cells infected with standard EHV-1 or DIP-enriched virus. These data also indicated that the UL2 polypeptide was more abundant in DIP-infected cells than in standard EHV-1-infected cells. Results from time course and pulse-chase analyses suggested that the UL2 polypeptide has a rapid turnover rate in DIP-infected cells.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bangham C. R., Kirkwood T. B. Defective interfering particles: effects in modulating virus growth and persistence. Virology. 1990 Dec;179(2):821–826. doi: 10.1016/0042-6822(90)90150-p. [DOI] [PubMed] [Google Scholar]
- Baumann R. P., Dauenhauer S. A., Caughman G. B., Staczek J., O'Callaghan D. J. Structure and genetic complexity of the genomes of herpesvirus defective-interfering particles associated with oncogenic transformation and persistent infection. J Virol. 1984 Apr;50(1):13–21. doi: 10.1128/jvi.50.1.13-21.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumann R. P., Staczek J., O'Callaghan D. J. Equine herpesvirus type 1 defective-interfering (DI) particle DNA structure: the central region of the inverted repeat is deleted from DI DNA. Virology. 1987 Jul;159(1):137–146. doi: 10.1016/0042-6822(87)90356-4. [DOI] [PubMed] [Google Scholar]
- Baumann R. P., Yalamanchili V. R., O'Callaghan D. J. Functional mapping and DNA sequence of an equine herpesvirus 1 origin of replication. J Virol. 1989 Mar;63(3):1275–1283. doi: 10.1128/jvi.63.3.1275-1283.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Block T., Jordan R., Farkas D. H., Hughes R. G., Jr Inhibition of transient gene expression with plasmids encoding herpes simplex virus type 1 UL55 and alpha genes. J Gen Virol. 1991 Jan;72(Pt 1):131–141. doi: 10.1099/0022-1317-72-1-131. [DOI] [PubMed] [Google Scholar]
- Campbell D. E., Kemp M. C., Perdue M. L., Randall C. C., Gentry G. A. Equine herpesvirus in vivo: cyclic production of a DNA density variant with repetitive sequences. Virology. 1976 Feb;69(2):737–750. doi: 10.1016/0042-6822(76)90502-x. [DOI] [PubMed] [Google Scholar]
- Cummings P. J., Rinaldo C. R., Jr Coevolution of virulent virus and resistant cells as a mechanism of persistence of herpes simplex virus type 1 in a human T lymphoblastoid cell line. J Gen Virol. 1989 Jan;70(Pt 1):97–106. doi: 10.1099/0022-1317-70-1-97. [DOI] [PubMed] [Google Scholar]
- Dauenhauer S. A., Robinson R. A., O'Callaghan D. J. Chronic production of defective-interfering particles by hamster embryo cultures of herpesvirus persistently infected and oncogenically transformed cells. J Gen Virol. 1982 May;60(Pt 1):1–14. doi: 10.1099/0022-1317-60-1-1. [DOI] [PubMed] [Google Scholar]
- Davison A. J., McGeoch D. J. Evolutionary comparisons of the S segments in the genomes of herpes simplex virus type 1 and varicella-zoster virus. J Gen Virol. 1986 Apr;67(Pt 4):597–611. doi: 10.1099/0022-1317-67-4-597. [DOI] [PubMed] [Google Scholar]
- Dohner D. E., Adams S. G., Gelb L. D. Varicella-zoster virus DNA from persistently infected cells contains novel tandem duplications. J Gen Virol. 1988 Sep;69(Pt 9):2229–2249. doi: 10.1099/0022-1317-69-9-2229. [DOI] [PubMed] [Google Scholar]
- Domke-Opitz I., Poberschin P., Mittnacht S., Kirchner H. Role of interferon in persistent infection of macrophages with herpes simplex virus. Virology. 1987 Aug;159(2):306–311. doi: 10.1016/0042-6822(87)90468-5. [DOI] [PubMed] [Google Scholar]
- Grundy F. J., Baumann R. P., O'Callaghan D. J. DNA sequence and comparative analyses of the equine herpesvirus type 1 immediate early gene. Virology. 1989 Sep;172(1):223–236. doi: 10.1016/0042-6822(89)90124-4. [DOI] [PubMed] [Google Scholar]
- Haeuptle M. T., Frank R., Dobberstein B. Translation arrest by oligodeoxynucleotides complementary to mRNA coding sequences yields polypeptides of predetermined length. Nucleic Acids Res. 1986 Feb 11;14(3):1427–1448. doi: 10.1093/nar/14.3.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harty R. N., Colle C. F., Grundy F. J., O'Callaghan D. J. Mapping the termini and intron of the spliced immediate-early transcript of equine herpesvirus 1. J Virol. 1989 Dec;63(12):5101–5110. doi: 10.1128/jvi.63.12.5101-5110.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harty R. N., O'Callaghan D. J. An early gene maps within and is 3' coterminal with the immediate-early gene of equine herpesvirus 1. J Virol. 1991 Jul;65(7):3829–3838. doi: 10.1128/jvi.65.7.3829-3838.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harty R. N., O'Callaghan D. J. Identification and expression of the UL1 gene product of equine herpesvirus 1. Virus Res. 1992 Sep 1;25(1-2):105–116. doi: 10.1016/0168-1702(92)90103-g. [DOI] [PubMed] [Google Scholar]
- Harty R. N., Yalamanchili R. R., O'Callaghan D. J. Transcriptional analysis of the UL1 gene of equine herpesvirus 1: a gene conserved in the genome of defective interfering particles. Virology. 1991 Aug;183(2):830–833. doi: 10.1016/0042-6822(91)91020-h. [DOI] [PubMed] [Google Scholar]
- Henry B. E., Newcomb W. W., O'Callaghan D. J. Biological and biochemical properties of defective interfering particles of equine herpesvirus type 1. Virology. 1979 Jan 30;92(2):495–506. doi: 10.1016/0042-6822(79)90152-1. [DOI] [PubMed] [Google Scholar]
- Holden V. R., Yalamanchili R. R., Harty R. N., O'Callaghan D. J. ICP22 homolog of equine herpesvirus 1: expression from early and late promoters. J Virol. 1992 Feb;66(2):664–673. doi: 10.1128/jvi.66.2.664-673.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holland J., Spindler K., Horodyski F., Grabau E., Nichol S., VandePol S. Rapid evolution of RNA genomes. Science. 1982 Mar 26;215(4540):1577–1585. doi: 10.1126/science.7041255. [DOI] [PubMed] [Google Scholar]
- Koerner T. J., Hill J. E., Myers A. M., Tzagoloff A. High-expression vectors with multiple cloning sites for construction of trpE fusion genes: pATH vectors. Methods Enzymol. 1991;194:477–490. doi: 10.1016/0076-6879(91)94036-c. [DOI] [PubMed] [Google Scholar]
- Levine M., Goldin A. L., Glorioso J. C. Persistence of herpes simplex virus genes in cells of neuronal origin. J Virol. 1980 Jul;35(1):203–210. doi: 10.1128/jvi.35.1.203-210.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGeoch D. J., Dalrymple M. A., Davison A. J., Dolan A., Frame M. C., McNab D., Perry L. J., Scott J. E., Taylor P. The complete DNA sequence of the long unique region in the genome of herpes simplex virus type 1. J Gen Virol. 1988 Jul;69(Pt 7):1531–1574. doi: 10.1099/0022-1317-69-7-1531. [DOI] [PubMed] [Google Scholar]
- O'Callaghan D. J., Hyde J. M., Gentry G. A., Randall C. C. Kinetics of viral deoxyribonucleic acid, protein, and infectious particle production and alterations in host macromolecular syntheses in equine abortion (herpes) virus-infected cells. J Virol. 1968 Aug;2(8):793–804. doi: 10.1128/jvi.2.8.793-804.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perdue M. L., Kemp M. C., Randall C. C., O'Callaghan D. J. Studies of the molecular anatomy of the L-M cell strain of equine herpes virus type 1: proteins of the nucleocapsid and intact virion. Virology. 1974 May;59(1):201–216. doi: 10.1016/0042-6822(74)90216-5. [DOI] [PubMed] [Google Scholar]
- Rigby P. W., Dieckmann M., Rhodes C., Berg P. Labeling deoxyribonucleic acid to high specific activity in vitro by nick translation with DNA polymerase I. J Mol Biol. 1977 Jun 15;113(1):237–251. doi: 10.1016/0022-2836(77)90052-3. [DOI] [PubMed] [Google Scholar]
- Robey W. G., Graham B. J., Harris C. L., Madden M. J., Pearson G. R., Vande Woude G. F. Persistent herpes simplex virus infections established in two Burkitt lymphoma derived cell lines. J Gen Virol. 1976 Jul;32(1):51–62. doi: 10.1099/0022-1317-32-1-51. [DOI] [PubMed] [Google Scholar]
- Robinson R. A., Tucker P. W., Dauenhauer S. A., O'Callaghan D. J. Molecular cloning of equine herpesvirus type 1 DNA: analysis of standard and defective viral genomes and viral sequences in oncogenically transformed cells. Proc Natl Acad Sci U S A. 1981 Nov;78(11):6684–6688. doi: 10.1073/pnas.78.11.6684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson R. A., Vance R. B., O'Callaghan D. J. Oncogenic transformation by by equine herpesviruses. II. Coestablishment of persistent infection and oncogenic transformation of hamster embryo cells by equine herpesvirus type 1 preparations enriched for defective interfering particles. J Virol. 1980 Oct;36(1):204–219. doi: 10.1128/jvi.36.1.204-219.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roux L., Simon A. E., Holland J. J. Effects of defective interfering viruses on virus replication and pathogenesis in vitro and in vivo. Adv Virus Res. 1991;40:181–211. doi: 10.1016/S0065-3527(08)60279-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith R. H., Caughman G. B., O'Callaghan D. J. Characterization of the regulatory functions of the equine herpesvirus 1 immediate-early gene product. J Virol. 1992 Feb;66(2):936–945. doi: 10.1128/jvi.66.2.936-945.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith R. H., Zhao Y., O'Callaghan D. J. The equine herpesvirus 1 (EHV-1) UL3 gene, an ICP27 homolog, is necessary for full activation of gene expression directed by an EHV-1 late promoter. J Virol. 1993 Feb;67(2):1105–1109. doi: 10.1128/jvi.67.2.1105-1109.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Telford E. A., Watson M. S., McBride K., Davison A. J. The DNA sequence of equine herpesvirus-1. Virology. 1992 Jul;189(1):304–316. doi: 10.1016/0042-6822(92)90706-u. [DOI] [PubMed] [Google Scholar]
- Thiele K., Mittnacht S., Kirchner H. Persistent replication of herpes simplex virus type 1 in JOK-1 cells. J Gen Virol. 1989 Jul;70(Pt 7):1907–1911. doi: 10.1099/0022-1317-70-7-1907. [DOI] [PubMed] [Google Scholar]
- Yalamanchili R. R., O'Callaghan D. J. Organization and function of the ORIs sequence in the genome of EHV-1 DI particles. Virology. 1990 Dec;179(2):867–870. doi: 10.1016/0042-6822(90)90157-m. [DOI] [PubMed] [Google Scholar]
- Yalamanchili R. R., O'Callaghan D. J. Sequence and organization of the genomic termini of equine herpesvirus type 1. Virus Res. 1990 Feb;15(2):149–161. doi: 10.1016/0168-1702(90)90005-v. [DOI] [PubMed] [Google Scholar]
- Yalamanchili R. R., Raengsakulrach B., Baumann R. P., O'Callaghan D. J. Identification of the site of recombination in the generation of the genome of DI particles of equine herpesvirus type 1. Virology. 1990 Apr;175(2):448–455. doi: 10.1016/0042-6822(90)90429-u. [DOI] [PubMed] [Google Scholar]
- Yalamanchili R. R., Raengsakulrach B., O'Callaghan D. J. Equine herpesvirus 1 sequence near the left terminus codes for two open reading frames. Virus Res. 1991 Mar;18(2-3):109–116. doi: 10.1016/0168-1702(91)90012-k. [DOI] [PubMed] [Google Scholar]
- Zhao Y., Holden V. R., Harty R. N., O'Callaghan D. J. Identification and transcriptional analyses of the UL3 and UL4 genes of equine herpesvirus 1, homologs of the ICP27 and glycoprotein K genes of herpes simplex virus. J Virol. 1992 Sep;66(9):5363–5372. doi: 10.1128/jvi.66.9.5363-5372.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]