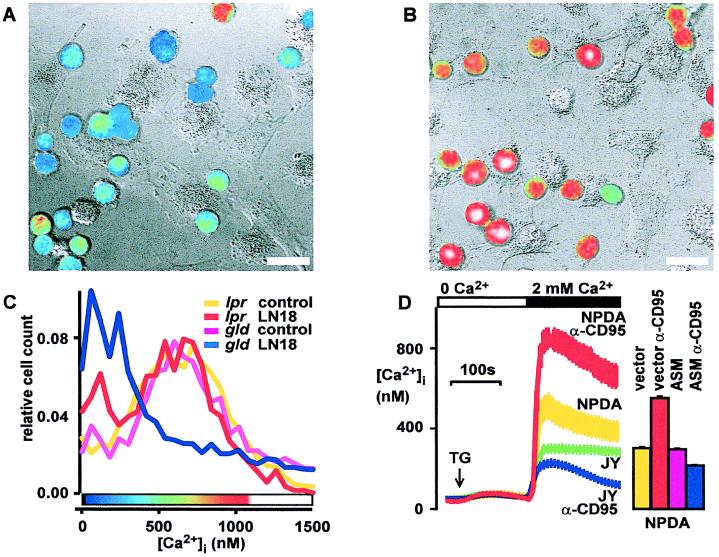
Figure 3.
CD95-induced block of store-operated Ca2+ entry requires CD95 and ASM. (A) Coculture with CD95L+ glioblastoma (LN18) blocks store-operated Ca2+ entry in T lymphocytes. Similar results were obtained with a second CD95L+ glioblastoma cell line (LN229). Fura-2 loaded Jurkat T cells were cocultured with CD95L+ glioblastoma cells (LN18) for 1.5 h and Ca2+ entry was stimulated with TG (1 μM for 10 min). Pseudocolored ratio images of lymphocyte fluorescence encode intracellular free Ca2+ concentrations according to the color bar shown in C. Fluorescence ratio images of lymphocytes were digitally overlaid on transmission images obtained simultaneously with differential interference contrast. (Bars = 20 μm.) (B) Neutralization of CD95L with a CD95-Fc-fusion protein (3 μg/ml) prevents block of Ca2+ entry in T cells following coculture with LN18 tumor. (C) Ca2+ block requires a functional CD95 receptor. In gld thymocytes, coculture blocks Ca2+ entry. In contrast, in CD95-defective lpr thymocytes, influx of Ca2+ is not blocked. Frequency distribution histograms of [Ca2+]i in single TG-stimulated thymocytes cocultured with LN18 tumor are shown. [Ca2+]i was measured in 104 cells by using flow cytometry. The color bar shows the pseudocolor scale for panels A and B. (D) ASM-deficient cells respond to CD95 stimulation (100 ng/ml anti-CD95 Ab for 1 h) with an increased Ca2+ plateau, whereas the [Ca2+]i rise evoked by TG in control cells is inhibited. Without CD95 triggering, the store-operated [Ca2+]i rise is higher in ASM-deficient cells (NPDA) compared with control (JY) cells. [Ca2+]i was measured with imaging microscopy in fura-2 loaded cells. Reconstitution of ASM into NPDA cells restores the Ca2+ block by CD95. Stimulated Ca2+ entry is increased after 1 h of CD95 stimulation in control-transfected NPDA cells. ASM transfection reverses this effect, leading to a block of Ca2+ entry by CD95. ASM transfection without CD95 stimulation has no effect on [Ca2+]i. Bars indicate mean [Ca2+]i levels 5 min after TG stimulation in 104 transfected NPDA cells measured by flow cytometry. Two independent experiments were performed.