Abstract
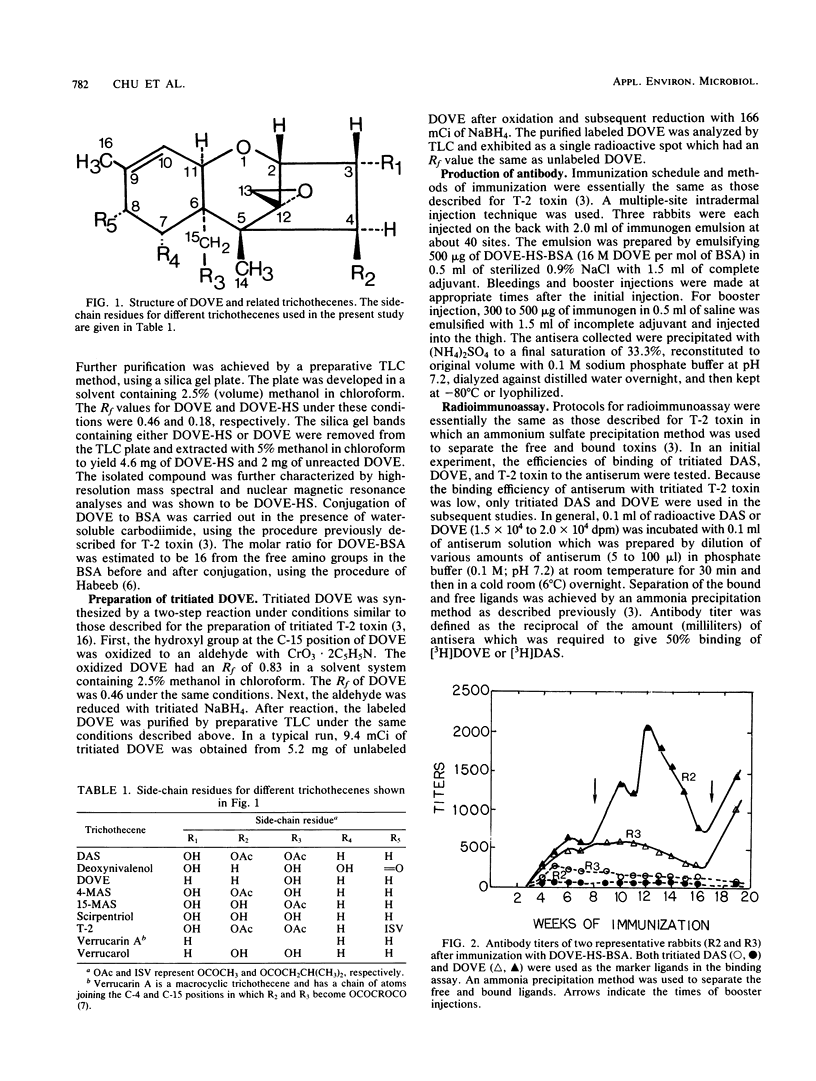
Immunization of rabbits with deoxyverrucarol (DOVE) conjugated to bovine serum albumin resulted in antibodies bound with either tritiated DOVE or diacetoxyscirpenol (DAS), but not with T-2 toxin. The affinity of antibodies with DOVE was found to be much higher than with DAS. When [3H] DOVE was used as a marking ligand in the competitive radioimmunoassay, the concentrations causing 50% inhibition of binding radioactivities by unlabeled DOVE, verrucarol, verrucarin A, and 4-monoacetoxyscirpenol were found to be 0.32, 1,070, 9,500, and 10,000 ng per assay, respectively. T-2 toxin, 15-monoacetoxyscirpenol, and deoxynivalenol gave less than 20% inhibition at 10 micrograms per assay. However, when [3H] DAS was used as the marking ligand, the concentrations causing 50% inhibition by DOVE, DAS, and verrucarol were found to be in the 50 to 60 ng per assay range. The antibodies are thus highly specific to DOVE rather than a common trichothecene backbone. The possible use of this antiserum for assay of macrocyclic trichothecenes is discussed.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Chu F. S., Grossman S., Wei R. D., Mirocha C. J. Production of antibody against T-2 toxin. Appl Environ Microbiol. 1979 Jan;37(1):104–108. doi: 10.1128/aem.37.1.104-108.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu F. S., Liang M. Y., Zhang G. S. Production and characterization of antibody against diacetoxyscirpenol. Appl Environ Microbiol. 1984 Oct;48(4):777–780. doi: 10.1128/aem.48.4.777-780.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fontelo P. A., Beheler J., Bunner D. L., Chu F. S. Detection of T-2 toxin by an improved radioimmunoassay. Appl Environ Microbiol. 1983 Feb;45(2):640–643. doi: 10.1128/aem.45.2.640-643.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habeeb A. F. Determination of free amino groups in proteins by trinitrobenzenesulfonic acid. Anal Biochem. 1966 Mar;14(3):328–336. doi: 10.1016/0003-2697(66)90275-2. [DOI] [PubMed] [Google Scholar]
- Jarvis B. B., Yatawara C. S., Greene S. L., Vrudhula V. M. Production of verrucarol. Appl Environ Microbiol. 1984 Sep;48(3):673–674. doi: 10.1128/aem.48.3.673-674.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S. C., Beery J. T., Chu F. S. Immunoperoxidase localization of T-2 toxin. Toxicol Appl Pharmacol. 1984 Feb;72(2):228–235. doi: 10.1016/0041-008x(84)90307-7. [DOI] [PubMed] [Google Scholar]
- Lee S., Chu F. S. Radioimmunoassay of T-2 toxin in biological fluids. J Assoc Off Anal Chem. 1981 May;64(3):684–688. [PubMed] [Google Scholar]
- Lee S., Chu F. S. Radioimmunoassay of T-2 toxin in corn and wheat. J Assoc Off Anal Chem. 1981 Jan;64(1):156–161. [PubMed] [Google Scholar]
- Peters H., Dierich M. P., Dose K. Enzyme-linked immunosorbent assay for detection of T-2 toxin. Hoppe Seylers Z Physiol Chem. 1982 Dec;363(12):1437–1441. doi: 10.1515/bchm2.1982.363.2.1437. [DOI] [PubMed] [Google Scholar]
- Wallace E. M., Pathre S. V., Mirocha C. J., Robison T. S., Fenton S. W. Synthesis of radiolabeled T-2 toxin. J Agric Food Chem. 1977 Jul-Aug;25(4):836–838. doi: 10.1021/jf60212a058. [DOI] [PubMed] [Google Scholar]
- Wei R., Strong F. M., Smalley E. B., Schnoes H. K. Chemical interconversion of T-2 and HT-2 toxins and related compounds. Biochem Biophys Res Commun. 1971 Oct 15;45(2):396–401. doi: 10.1016/0006-291x(71)90832-1. [DOI] [PubMed] [Google Scholar]


