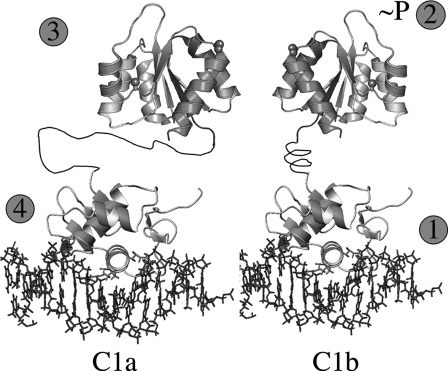
FIGURE 12.
Sequence of events for OmpR activation and DNA binding. Step1, OmpR binds as a monomer to the high affinity C1b site located 3′ to its upstream half-site C1a. Step 2, conformational change occurs in the N-terminal receiver domain as a consequence of the C terminus binding to DNA. This promotes phosphorylation of Asp55 in the receiver domain. Step 3, as a consequence of phosphorylation, the receiver domains form symmetric dimers. Step 4, dimerization brings the second OmpRC domain to its lower affinity site, promoting DNA binding by the second OmpRC. If the orientation of C1b is reversed, the dimer still forms, presumably because the longer, more flexible linker enables OmpRC to bind.