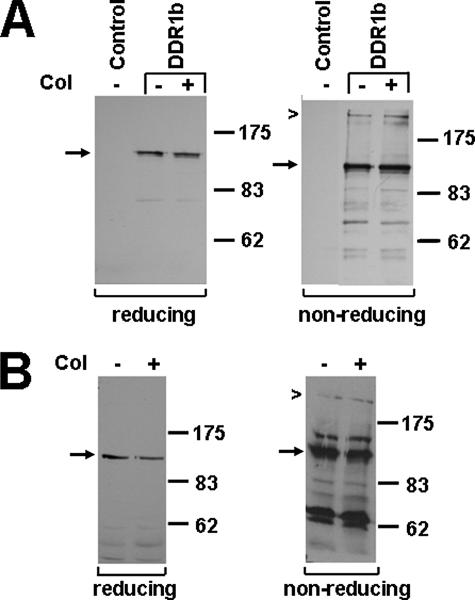
FIGURE 2.
DDR1b forms covalent dimers. A, 293 cells were transiently transfected with DDR1b or an empty plasmid (control) and stimulated with type I collagen (col) for 90 min. Using an antibody specific for the C terminus of DDR1, Western blot analysis of gels run under reducing conditions (left panel) detected DDR1b as a monomeric protein (arrow), whereas under nonreducing conditions (right panel), DDR1b was present as both a monomer (arrow) and a dimer (chevron). B, T-47D cells that endogenously express DDR1b were stimulated with collagen for 18 h and lysates analyzed by Western blotting under reducing (left panel) and nonreducing conditions (right panel). The experiments were performed three times, and representative data are shown.