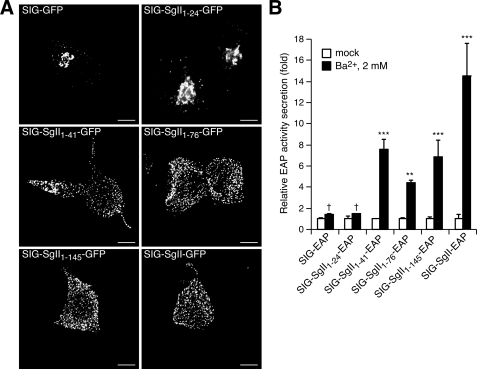
FIGURE 7.
Trafficking of progressively truncated N-terminal domains of SgII fused to GFP or EAP in PC12 cells. Cells transfected with expression plasmids encoding the indicated photoproteins were processed for deconvolution microscopy (A). A transition from a punctate/vesicular distribution (e.g. SIG-SgII-GFP) to a juxtanuclear localization (e.g. SIG-GFP) is seen as the N-terminal domain of SgII is reduced from +41 to +24 residues. B, secretagogue-evoked release of SgII domain-EAP chimeras. PC12 cells expressing the indicated EAP fusion proteins were exposed for 15 min to secretion medium alone (mock) or to 2 mm Ba2+. The enzymatic activity of EAP chimeras was assayed in the culture supernatant and cell lysate, and relative secretion was determined as described in the legend of Fig. 4. Values are given as the means ± S.E. of triplicate determinations. †, p > 0.05; **, p < 0.01; ***, p < 0.001 as compared with control (mock), ANOVA with Dunnett's post test. Scale bar, 5 μm.