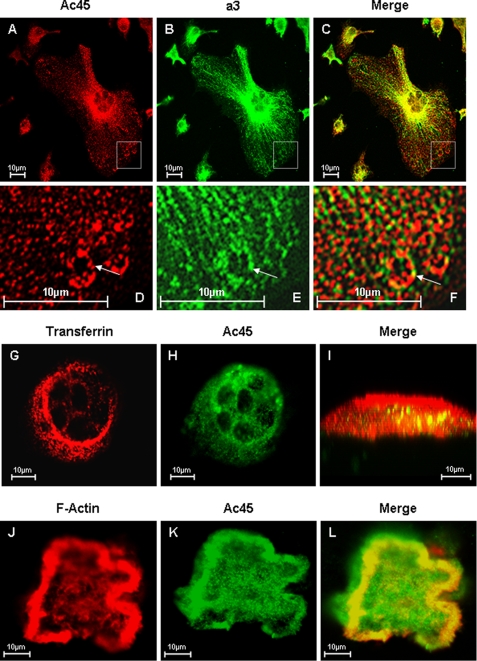
FIGURE 3.
Confocal microscopy analysis of Ac45 co-localization with a3 in OCLs. A–E, osteoclasts cultured on glass coverslips were fixed and permeabilized in 0.1% Triton X-100. The cells were immunostained with primary rabbit Anti-Ac45 and guinea pig anti-a3 for 2 h followed by secondary goat anti-rabbit Alexa Fluor 546 (A) and fluorescein isothiocyanate-conjugated anti-guinea pig (B) for 45 min, respectively. Co-localization was examined following the overlaying of the Ac45 and a3 signals (C). D–F, higher magnification of the boxed area. G–I, osteoclasts seeded onto bone slices for 3 days were incubated with Alexa Fluor 546-conjugated transferrin (G) for 30 min followed by fixation and permeabilization. The cells were immunostained with an anti-Ac45 antibody (H) for 2 h and then with secondary goat anti-rabbit Alexa Fluor 488 for 45 min before confocal analysis. Co-localization between Ac45 and transferrin filled endosomal vesicles were observed in z-x or vertical section imaging (I). J–L, confocal microscopy analysis of Ac45 protein localization in resorbing osteoclasts. Following 3-day culture on bone slices, osteoclasts were fixed and permeabilized in 0.1% Triton X-100. The cells were again immunostained for with an anti-Ac45 antibody (J) and secondary goat anti-rabbit Alexa Fluor 488. F-actin was stained with rhodamine-conjugated phalloidin (K). Ac45 was observed to extensively co-localize with F-actin surrounding the resorption lacunae (L).