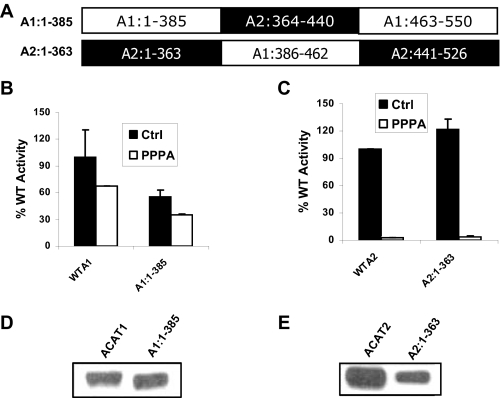
FIGURE 1.
PPPA-sensitive site of ACAT2 is located outside the putative active site domain of the enzyme. A, primary structure of the chimeric proteins termed A1:1–385 and A2:1–363 are indicated where sequences from ACAT1 are in open boxes and sequences from ACAT2 are in filled boxes. B, kinetic assay data for WTA1 and A1:1–385. AC29 cells were transfected with the cDNA encoding WTA1 and A1:1–385 proteins. 72 h post-transfection cells were incubated with either vehicle (Me2SO) or 5μm PPPA for 30 min at 37 °C. There after cells were pulse-labeled with 1 μCi of [3H]oleic acid for 2 h. The incorporation of [3H]oleic acid into cellular CE pool was measured as the determinant of the enzymatic activity of the respective proteins. Background activity was obtained by a parallel kinetic assay where cells were transfected with an empty vector. All activities were corrected by background subtraction and were normalized against the control (Ctrl) WT activity. Data represent the mean ± S.E. for n = 2. This experiment was repeated three times with similar results. C, kinetic assay data for of WTA2 and A2:1–363. The assay was performed essentially as described above, and data are presented as above. D, PNS made from cells transfected with WTA1 and A1:1–385 cDNAs were subjected to immunoblot using affinity-purified ACAT1 antibody as described under “Experimental Procedures.” E, PNS, made from the cells transfected with WTA2 and A2:1–363 cDNAs, were subjected to Western blot analysis as described above.