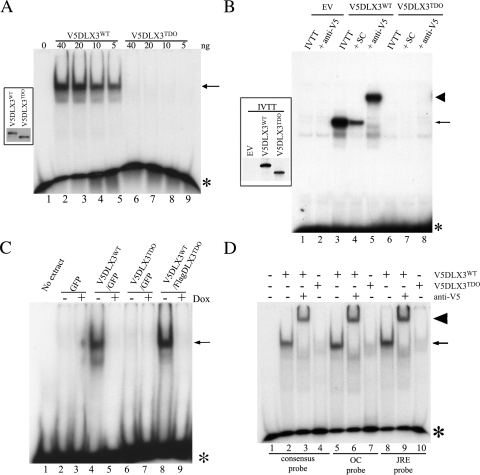
FIGURE 2.
DNA binding capacity of DLX3WT and DLX3TDO. EMSA were performed to compare the capacity of DLX3WT and DLX3TDO to bind DNA. A, recombinant V5DLX3WT and V5DLX3TDO were produced in E. coli, purified, and different amounts of protein were analyzed in EMSA using the Dlx3 consensus probe (GGGGGATAATTGCTGG). The inset on the left-hand side shows a Western blot analysis of both recombinant proteins using anti-V5 antibody. B, V5DLX3WT and V5DLX3TDO were in vitro transcribed/translated (IVTT) from pet15b-V5DLX3WT and pet15b-V5DLX3TDO (in vitro transcribed/translated, reticulocyte lysate system) and analyzed by EMSA using a probe containing a Dlx3 binding site previously identified in the Runx2 promoter. Empty vector (pet15b, EV) was used as a control. Excess unlabeled probe were used for self-competition (SC) and anti-V5 antibody was used for supershift assays. Asterisks, free radiolabeled probe; arrows, protein-DNA complexes; arrowheads, antibody-protein-DNA complexes. C, Saos2-Tet-Off cells transfected with pBi-GFP, pBi-V5DLX3WT/GFP, pBi-V5DLX3TDO/GFP, or pBi-V5DLX3WT/FlagDLX3TDO were grown with or without doxycycline for 48 h. Nuclear extracts were harvested and EMSA was performed using the Dlx3 consensus probe. D, recombinant V5DLX3WT and V5DLX3TDO were analyzed by EMSA using different probes known to bind Dlx3: a probe containing the Dlx3 consensus binding site determined by SELEX (GGGGGATAATTGCTGG), a probe containing the Dlx3 binding site identified in the osteocalcin (OC) promoter (GGGCCCCCAATTAGTCCTCCC), and a probe containing the junctional regulatory element (JRE) present in the promoter of the glycoprotein hormone α subunit expressed in the placenta. Anti-V5 antibody was used for supershift assays.