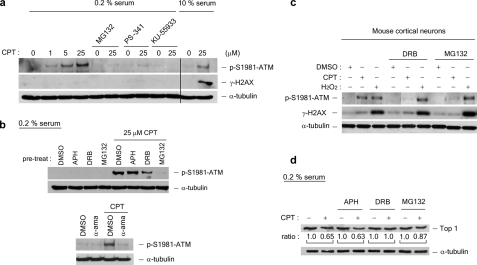
FIGURE 2.
CPT-induced ATM autophosphorylation is transcription- and proteasome-dependent in quiescent WI-38 cells. WI-38 cells cultured in DMEM supplemented with 0.2% serum were treated with CPT (1, 5, or 25 μm) for 1 h in the presence and absence of various inhibitors. A proteasome inhibitor (2 μm MG132 or 1 μm PS-341) was added 30 min prior to CPT addition. The ATM inhibitor KU-55933 (10 μg/ml) was added 60 min prior to CPT addition. The cell lysates were immunoblotted with antibodies as indicated. As controls (the two right-most lanes in a), lysates from proliferating WI-38 cells (cultured in 10% serum) were immunoblotted. b, upper panels, WI-38 cells cultured in DMEM supplemented with 0.2% serum were preincubated with the replication inhibitor APH (10 μm) or the transcription inhibitor DRB (50 μm) for 30 min prior to CPT addition. Co-treatment with CPT was for 1 h. The cell lysates were immunoblotted with antibodies as indicated. b, lower panels, the same as in b (upper panels), except that the RNA polymerase inhibitorα-amanitin (5 μg/ml) was used during co-treatment with CPT. c, post-mitotic mouse cortical neurons were treated with CPT (20 μm) or H2O2 (100 μm) in the presence of MG132 (4 μm) or DRB (150 μm) for 1 h. The cell lysates were immunoblotted. d, Top1 down-regulation in WI-38 cells cultured in DMEM supplemented with 0.2% serum. The cells were pretreated with different inhibitors (APH, DRB, or MG132) for 30 min, followed by co-treatment with CPT (25 μm) for 3 h. Following a 30-min incubation in fresh medium (without inhibitors or CPT), the cells were lysed by an alkaline lysis procedure, and the cell lysates were subject to staphylococcal nuclease S7 treatment (see “Experimental Procedures”). The cell lysates were then analyzed by immunoblotting using hTop1 antisera. The relative band intensities (expressed in ratios; the relative band intensities of samples without CPT treatment were taken as 1.0) were quantified by KODAK one-dimensional image analysis software. DMSO, dimethyl sulfoxide.