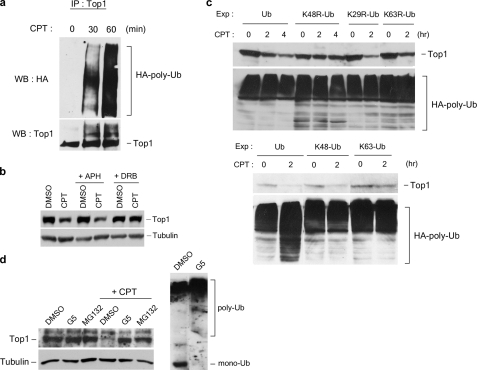
FIGURE 5.
CPT-induced Top1 down-regulation is dependent on the assembly of K48-linked polyubiquitin chains. a, HeLa cells were transfected with the HA-Ub expression plasmid. Transfection, CPT treatment, and immunoprecipitation (IP) were performed as described under “Experimental Procedures.” b, HeLa cells were pretreated with 10 μm APH or 150 μm DRB for 30 min, followed by co-treatment with 25 μm CPT for 3 h. Top1 down-regulation was analyzed as described under “Experimental Procedures.” c, HeLa cells were transfected with various plasmids to overexpress HA-tagged wild type and mutant ubiquitin proteins. Transfection, CPT treatment (25 μm), and Top1 down-regulation assay were performed as described under “Experimental Procedures.” Ub, wild type ubiquitin; K48R-Ub, K48R mutant ubiquitin; K29R-Ub, K29R mutant ubiquitin; K63R-Ub, K63R mutant ubiquitin; K48-Ub, K48-only ubiquitin (all lysine residues were mutated to arginine residues except Lys-48); K63-only Ub (all lysine residues were mutated to arginine residues except Lys-63). The amount of protein-HA-Ub conjugates in cells transfected with each construct was also determined by immunoblotting using anti-HA antibody (lower panels). d, Top1 degradation requires ubiquitin and proteasome. HeLa cells were pretreated with 5 μm MG132 or 5 μm G5 for 30 min, followed by co-treatment with 25 μm CPT for 3 h. Top1 down-regulation was determined by immunoblotting of cell lysates with anti-hTop1 antibody. Cell lysates (from control cells and G5-treated cells) were also immunoblotted with anti-ubiquitin antibody to assess the levels of free ubiquitin (mono-Ub) and ubiquitin-protein conjugates (poly-Ub). WB, Western blot; DMSO, dimethyl sulfoxide.