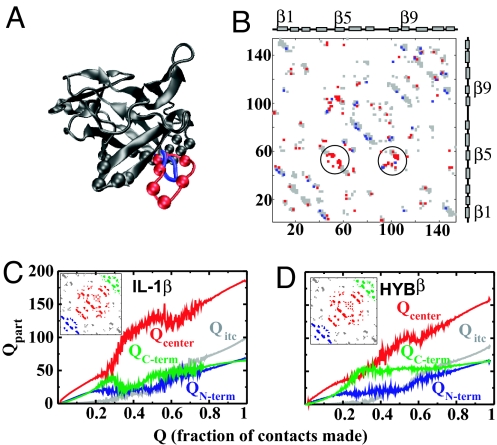
Fig. 3.
A comparison of IL-1β and HYBβ. (A) Aligned structures of HYBβ and IL-1β. The gray regions are common to both proteins. The differences are in red (IL-1β: the β-bulge) and blue (HYBβ: tight β-turn). The gray and red spheres depict the Cα atoms of the residues in the B-site. (B) Contact maps of IL-1β (422 contacts) and HYBβ (384 contacts). The map is symmetric about the diagonal, and both triangles are shown for clarity. Gray contacts are common to both proteins. The red contacts are present only in IL-1β and the blue ones only in HYBβ. The circled contacts involve the β-bulge. The x and y axes give residue numbers, and the index is of the larger protein, here, IL-1β. (C and D) How different parts of the protein fold. The Insets show the different regions into which the two proteins are partitioned. The color of the parts matches the color of the Qparts. (C) IL-1β. The initial rise and fall of the green QC-term denotes the backtracking. The red Qcenter shows a marked increase with a reduction in the structure of the green region. (D) HYBβ. The C-terminal region forms early and stays formed. The red region increases almost linearly with Q.