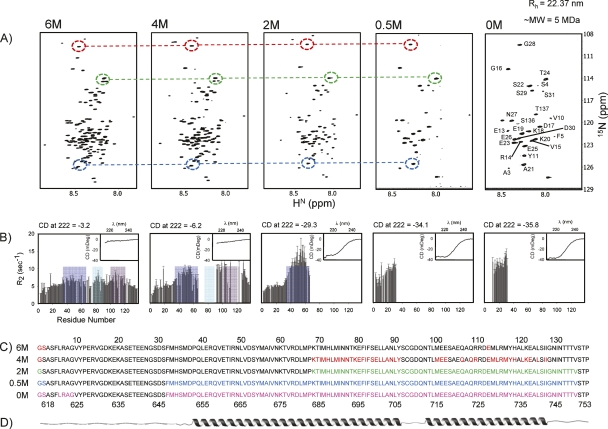
Figure 2.
Summary of the sequence of events that occur during the Gdn-HCl titration of GED as probed by NMR and CD. (A) 1H-15N HSQC spectra of GED at 800 MHz at varying Gdn-HCl concentrations; assignments at 0 M Gdn-HCl are marked; dashed circles and lines illustrate the similarity of the peak positions; there is a progressive disappearance of peaks as we proceed toward the folded state. (B) 15N transverse relaxation rates (R 2) measured at the corresponding Gdn-HCl concentrations as in A; blue, green, and purple patches correspond to the B, C, and D domains (see text) of restricted motion/conformational exchange; there is a progressive increase in the R 2 values followed by lack of data (due to peak disappearance in the HSQC spectra) in these domains; insets show far-UV CD spectra at the corresponding guanidine concentrations; ellipticity at 222 nm is shown at the top of each panel to indicate helicity changes; these are used to calculate molar ellipticity values and thereby the helical contents (see text). (C) Residues for which peaks disappeared in the HSQC spectra at different guanidine concentrations are marked in color on the sequence of the protein; residue numbers at the top are as per recombinant GED and those at the bottom are as per dynamin. (D) Predicted secondary structure along the sequence.