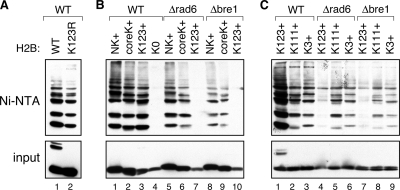
Figure 2.
There are at least two distinct pathways of H2B polyubiquitylation. Ubiquitylation of the indicated HA-tagged H2B mutants was measured as described in the legend to Figure 1, except that H2B–HA was expressed from a centromeric plasmid in strain BY4742. (A) The K123R H2B mutant is polyubiquitylated. (B) Ubiquitylation at lysine 123 depends exclusively on Rad6–Bre1. The ubiquitylation status of four different H2B mutants—NK+, coreK+, K123+, and K0 (described in the text)—was determined in either WT cells (lanes 1–4), or congenic strains in which RAD6 (FGY3; lanes 5–7) or BRE1 (FGY4; lanes 8–10) were deleted. (C) H2B proteins carrying single lysine residues are polyubiquitylated. The ubiquitylation status of K123+, K111+, and K3+ H2B mutants was examined in WT (lanes 1–3), Δrad6 (lanes 4–6), and Δbre1 (lanes 7–9) cells. Note that a more sensitive form of detection was used in the experiment in C than in B, because of the lower absolute signal from the single lysine K111+ and K3+ mutants. For this reason, monoubiquitylated K123+ H2B is visible in the input in C (cf. lanes in B with lane 1 in C).