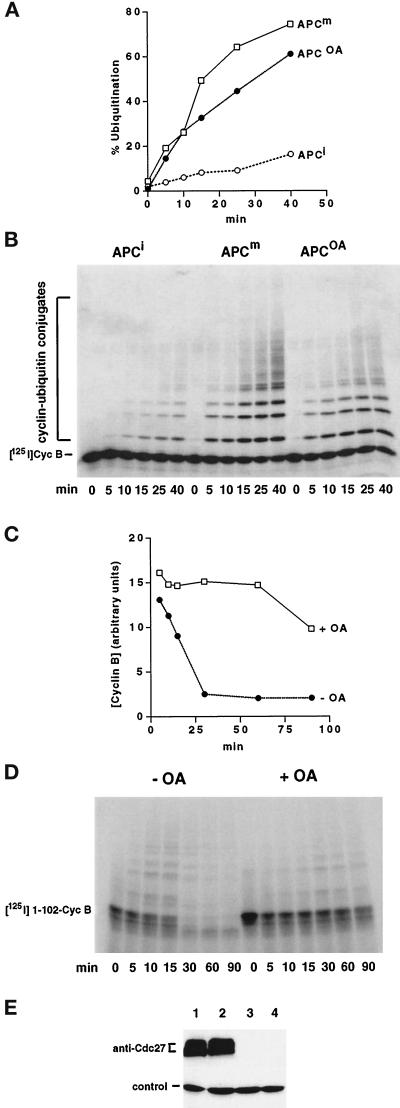
Figure 3.
OA does not efficiently block the mitotic activation of the APC, but activates an IMP. (A and B) Time course showing the ability of different APC immunoprecipitates to ubiquitinate 125I-labeled-cyclin B 13–110 (Cyc B) in a reconstituted system containing purified E1, UBC4, and UBCx. APC was isolated from an interphase extract (APCi), from a mitotic Δ90 extract (APCm), and from an extract treated simultaneously with Δ90 and 1 μM OA (APCOA). Samples were analyzed by SDS-PAGE and phosphorimaging (B), and the ubiquitination activities were expressed as percentage of cyclin B converted into conjugates (A). (C and D) The stability of 125I-labeled cyclin B1 1–102 (1–102-CycB) was analyzed in APC-depleted extracts supplemented with a mitotic APC fraction. Interphase extracts were either treated with Δ90 alone (−OA) or simultaneously with Δ90 and 1 μM OA (+OA) for 45 min before the APC depletion and reconstitution. Samples were analyzed as above (D), and cyclin B levels were quantitated (C). (E) Cdc27 immunoblot of the extracts used in C and D to control for the immunodepletion of APC. Lanes 1 and 3, Δ90 extract before and after depletion with Cdc27 antibodies, respectively; lanes 2 and 4, extract treated with Δ90 and OA at time zero, before and after Cdc27 immunodepletion, respectively. A protein band from a different portion of the same blot, which nonspecifically cross-reacts with Cdc27 antisera, is shown as a loading control.