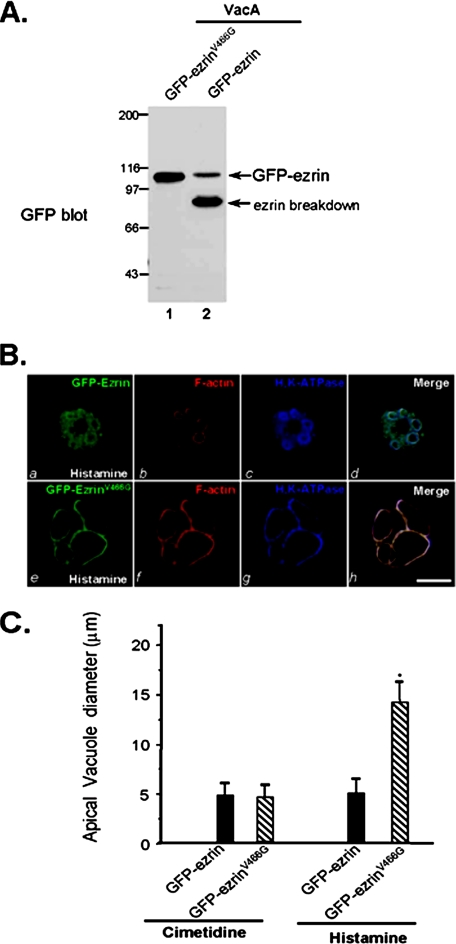
FIGURE 5.
Block of calpain-mediated ezrin hydrolysis rescues VacA-induced inhibition of acid secretion. A, aliquots of cultured parietal cells transiently transfected to express wild type GFP-ezrin and GFP-ezrinV466G mutant. Thirty-six hours after the transfection, the parietal cells were incubated with activated VacA (5 μg/ml) and boiled VacA in the presence of 1.8 mm Ca2+ for 20 min prior to being solubilized in SDS-PAGE sample buffer. Equivalent amounts of proteins from these samples were applied to SDS-PAGE and subsequently transblotted onto nitrocellulose membrane. The blot was probed with an anti-GFP antibody using an ECL kit (Pierce). Note that wild type GFP-ezrin but not GFP-ezrinV466G is hydrolyzed in VacA-treated glandular cells. B, cultured parietal cells transiently transfected to express wild type GFP-ezrin and GFP-ezrinV466G mutant. Thirty-six hours after the transfection, the parietal cells were incubated with activated VacA (10 μg/ml) and 100 mm histamine plus 50 mm IBMX in the presence of 1.8 mm Ca2+ for 20 min. Treated cells were fixed and stained for ezrin, F-actin, and H,K-ATPase. Note that the apical vacuoles are dilated in the GFP-ezrinV466G-expressing cells but not wild type GFP-ezrin-expressing cells. Bar, 10 μm. C, 35 parietal cells positively expressing GFP-ezrin (wild type and ezrinV466G mutant) and stimulated parietal cells were surveyed for apical vacuole diameters in the same focal plane as described in Fig. 4D. Data are expressed as means ± S.E.