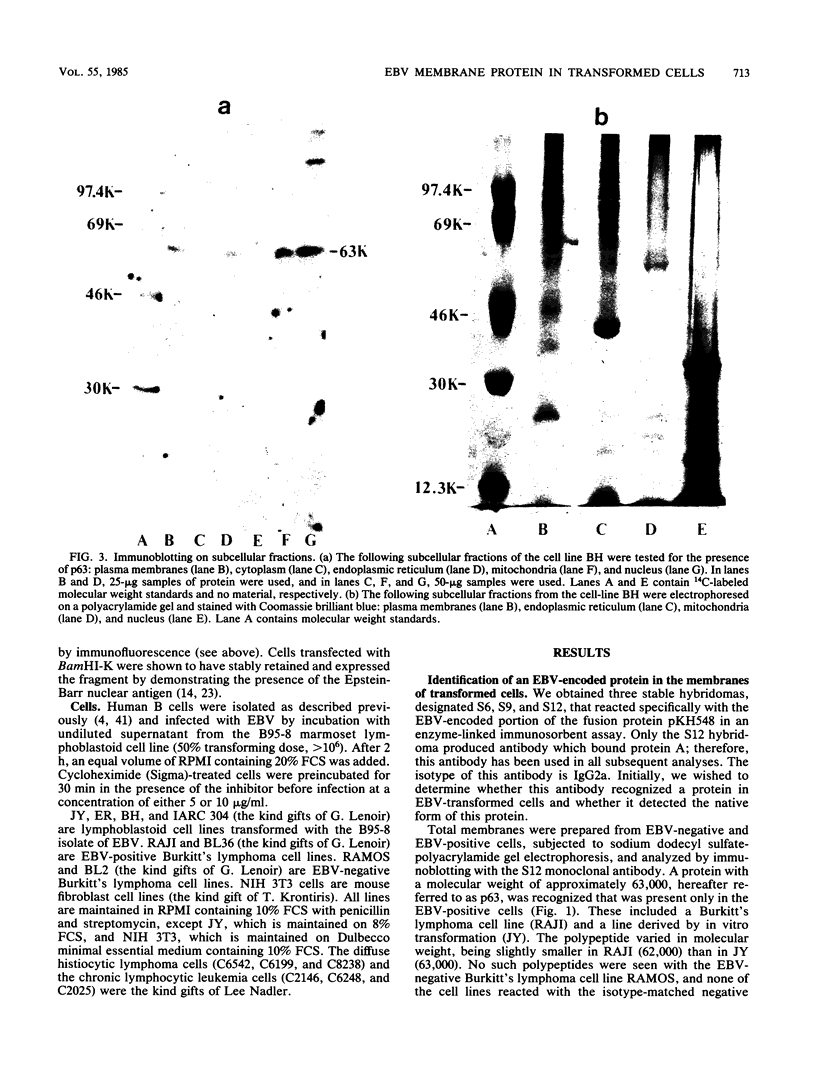
Abstract
We have developed monoclonal antibodies to a 63,000-molecular-weight protein (p63) which is the product of the most abundant messenger RNA in Epstein-Barr virus-transformed cells and shown that the protein is associated specifically with plasma membranes. It was also found to be associated with the other membrane fractions and was found in all Epstein-Barr virus-transformed cells tested. In addition, p63 was present in virions, resulting in transient, early appearance in newly infected cells. Newly synthesized p63 was detected at the time cells underwent blast transformation (48 to 72 h postinfection). The possible role of this protein in transformation and as a target for cell-mediated cytotoxicity is discussed.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baer R., Bankier A. T., Biggin M. D., Deininger P. L., Farrell P. J., Gibson T. J., Hatfull G., Hudson G. S., Satchwell S. C., Séguin C. DNA sequence and expression of the B95-8 Epstein-Barr virus genome. Nature. 1984 Jul 19;310(5974):207–211. doi: 10.1038/310207a0. [DOI] [PubMed] [Google Scholar]
- Bar R. S., DeLor C. J., Clausen K. P., Hurtubise P., Henle W., Hewetson J. F. Fatal infectious mononucleosis in a family. N Engl J Med. 1974 Feb 14;290(7):363–367. doi: 10.1056/NEJM197402142900704. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Crawford D. H., Epstein M. A., Bornkamm G. W., Achong B. G., Finerty S., Thompson J. L. Biological and biochemical observations on isolates of EB virus from the malignant epithelial cells of two nasopharyngeal carcinomas. Int J Cancer. 1979 Sep 15;24(3):294–302. doi: 10.1002/ijc.2910240305. [DOI] [PubMed] [Google Scholar]
- Crumpton M. J., Snary D. Preparation and properties of lymphocyte plasma membrane. Contemp Top Mol Immunol. 1974;3:27–56. doi: 10.1007/978-1-4684-2838-4_2. [DOI] [PubMed] [Google Scholar]
- Dambaugh T., Beisel C., Hummel M., King W., Fennewald S., Cheung A., Heller M., Raab-Traub N., Kieff E. Epstein-Barr virus (B95-8) DNA VII: molecular cloning and detailed mapping. Proc Natl Acad Sci U S A. 1980 May;77(5):2999–3003. doi: 10.1073/pnas.77.5.2999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Blas A. L., Cherwinski H. M. Detection of antigens on nitrocellulose paper immunoblots with monoclonal antibodies. Anal Biochem. 1983 Aug;133(1):214–219. doi: 10.1016/0003-2697(83)90245-2. [DOI] [PubMed] [Google Scholar]
- Dolyniuk M., Pritchett R., Kieff E. Proteins of Epstein-Barr virus. I. Analysis of the polypeptides of purified enveloped Epstein-Barr virus. J Virol. 1976 Mar;17(3):935–949. doi: 10.1128/jvi.17.3.935-949.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edson C. M., Thorley-Lawson D. A. Epstein-Barr virus membrane antigens: characterization, distribution, and strain differences. J Virol. 1981 Jul;39(1):172–184. doi: 10.1128/jvi.39.1.172-184.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Einhorn L., Ernberg I. Induction of EBNA precedes the first cellular S-phase after EBV-infection of human lymphocytes. Int J Cancer. 1978 Feb 15;21(2):157–160. doi: 10.1002/ijc.2910210205. [DOI] [PubMed] [Google Scholar]
- Fennewald S., van Santen V., Kieff E. Nucleotide sequence of an mRNA transcribed in latent growth-transforming virus infection indicates that it may encode a membrane protein. J Virol. 1984 Aug;51(2):411–419. doi: 10.1128/jvi.51.2.411-419.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischer D. K., Robert M. F., Shedd D., Summers W. P., Robinson J. E., Wolak J., Stefano J. E., Miller G. Identification of Epstein-Barr nuclear antigen polypeptide in mouse and monkey cells after gene transfer with a cloned 2.9-kilobase-pair subfragment of the genome. Proc Natl Acad Sci U S A. 1984 Jan;81(1):43–47. doi: 10.1073/pnas.81.1.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foung S. K., Sasaki D. T., Grumet F. C., Engleman E. G. Production of functional human T-T hybridomas in selection medium lacking aminopterin and thymidine. Proc Natl Acad Sci U S A. 1982 Dec;79(23):7484–7488. doi: 10.1073/pnas.79.23.7484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerper P., Whang-Peng J., Monroe J. H. Transformation and chromosome changes induced by Epstein-Barr virus in normal human leukocyte cultures. Proc Natl Acad Sci U S A. 1969 Jul;63(3):740–747. doi: 10.1073/pnas.63.3.740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham F. L., van der Eb A. J. A new technique for the assay of infectivity of human adenovirus 5 DNA. Virology. 1973 Apr;52(2):456–467. doi: 10.1016/0042-6822(73)90341-3. [DOI] [PubMed] [Google Scholar]
- Henderson E., Heston L., Grogan E., Miller G. Radiobiological inactivation of Epstein-Barr virus. J Virol. 1978 Jan;25(1):51–59. doi: 10.1128/jvi.25.1.51-59.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henle G., Henle W., Clifford P., Diehl V., Kafuko G. W., Kirya B. G., Klein G., Morrow R. H., Munube G. M., Pike P. Antibodies to Epstein-Barr virus in Burkitt's lymphoma and control groups. J Natl Cancer Inst. 1969 Nov;43(5):1147–1157. [PubMed] [Google Scholar]
- Henle W., Diehl V., Kohn G., Zur Hausen H., Henle G. Herpes-type virus and chromosome marker in normal leukocytes after growth with irradiated Burkitt cells. Science. 1967 Sep 1;157(3792):1064–1065. doi: 10.1126/science.157.3792.1064. [DOI] [PubMed] [Google Scholar]
- Henle W., Henle G., Zajac B. A., Pearson G., Waubke R., Scriba M. Differential reactivity of human serums with early antigens induced by Epstein-Barr virus. Science. 1970 Jul 10;169(3941):188–190. doi: 10.1126/science.169.3941.188. [DOI] [PubMed] [Google Scholar]
- Hennessy K., Fennewald S., Hummel M., Cole T., Kieff E. A membrane protein encoded by Epstein-Barr virus in latent growth-transforming infection. Proc Natl Acad Sci U S A. 1984 Nov;81(22):7207–7211. doi: 10.1073/pnas.81.22.7207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennessy K., Kieff E. One of two Epstein-Barr virus nuclear antigens contains a glycine-alanine copolymer domain. Proc Natl Acad Sci U S A. 1983 Sep;80(18):5665–5669. doi: 10.1073/pnas.80.18.5665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson G. S., Farrell P. J., Barrell B. G. Two related but differentially expressed potential membrane proteins encoded by the EcoRI Dhet region of Epstein-Barr virus B95-8. J Virol. 1985 Feb;53(2):528–535. doi: 10.1128/jvi.53.2.528-535.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King W., Thomas-Powell A. L., Raab-Traub N., Hawke M., Kieff E. Epstein-Barr virus RNA. V. Viral RNA in a restringently infected, growth-transformed cell line. J Virol. 1980 Nov;36(2):506–518. doi: 10.1128/jvi.36.2.506-518.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Köhler G., Milstein C. Derivation of specific antibody-producing tissue culture and tumor lines by cell fusion. Eur J Immunol. 1976 Jul;6(7):511–519. doi: 10.1002/eji.1830060713. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Land H., Parada L. F., Weinberg R. A. Cellular oncogenes and multistep carcinogenesis. Science. 1983 Nov 18;222(4625):771–778. doi: 10.1126/science.6356358. [DOI] [PubMed] [Google Scholar]
- Levy J. A., Henle G. Indirect immunofluorescence tests with sera from African children and cultured Burkitt lymphoma cells. J Bacteriol. 1966 Jul;92(1):275–276. doi: 10.1128/jb.92.1.275-276.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menezes J., Jondal M., Leibold W., Dorval G. Epstein-Barr virus interactions with human lymphocyte subpopulations: virus adsorption, kinetics of expression of Epstein-Barr virus-associated nuclear antigen, and lymphocyte transformation. Infect Immun. 1976 Feb;13(2):303–310. doi: 10.1128/iai.13.2.303-310.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misko I. S., Moss D. J., Pope J. H. HLA antigen-related restriction of T lymphocyte cytotoxicity to Epstein-Barr virus. Proc Natl Acad Sci U S A. 1980 Jul;77(7):4247–4250. doi: 10.1073/pnas.77.7.4247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss D. J., Burrows S. R., Parsons P. G. Calcium concentration defines two stages in transformation of lymphocytes by Epstein-Barr virus. Int J Cancer. 1984 May 15;33(5):587–590. doi: 10.1002/ijc.2910330507. [DOI] [PubMed] [Google Scholar]
- Old L. J., Boyse E. A., Oettgen H. F., Harven E. D., Geering G., Williamson B., Clifford P. Precipitating antibody in human serum to an antigen present in cultured burkitt's lymphoma cells. Proc Natl Acad Sci U S A. 1966 Dec;56(6):1699–1704. doi: 10.1073/pnas.56.6.1699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pope J. H., Horne M. K., Scott W. Identification of the filtrable leukocyte-transforming factor of QIMR-WIL cells as herpes-like virus. Int J Cancer. 1969 May 15;4(3):255–260. doi: 10.1002/ijc.2910040302. [DOI] [PubMed] [Google Scholar]
- Powell A. L., King W., Kieff E. Epstein-Barr virus-specific RNA. III. Mapping of DNA encoding viral RNA in restringent infection. J Virol. 1979 Jan;29(1):261–274. doi: 10.1128/jvi.29.1.261-274.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson J., Smith D. Infection of human B lymphocytes with high multiplicities of Epstein-Barr virus: kinetics of EBNA expression, cellular DNA synthesis, and mitosis. Virology. 1981 Mar;109(2):336–343. doi: 10.1016/0042-6822(81)90504-3. [DOI] [PubMed] [Google Scholar]
- Schneider U., zur Hausen H. Epstein-Barr virus-induced transformation of human leukocytes after cell fractionation. Int J Cancer. 1975 Jan 15;15(1):59–66. doi: 10.1002/ijc.2910150108. [DOI] [PubMed] [Google Scholar]
- Slovin S. F., Schooley R. T., Thorley-Lawson D. A. Analysis of cellular immune response to EBV by using cloned T cell lines. J Immunol. 1983 May;130(5):2127–2132. [PubMed] [Google Scholar]
- Takada K., Osato T. Analysis of the transformation of human lymphocytes by Epstein-Barr virus. I. Sequential occurrence from the virus-determined nuclear antigen synthesis, to blastogenesis, to DNA synthesis. Intervirology. 1979;11(1):30–39. doi: 10.1159/000149009. [DOI] [PubMed] [Google Scholar]
- Tanaka Y., Sugamura K., Hinuma Y. Heterogeneity of allogeneic restriction of human cytotoxic T cell clones specific for Epstein Barr virus. J Immunol. 1982 Mar;128(3):1241–1245. [PubMed] [Google Scholar]
- Thorley-Lawson D. A., Chess L., Strominger J. L. Suppression of in vitro Epstein-Barr virus infection. A new role for adult human T lymphocytes. J Exp Med. 1977 Aug 1;146(2):495–508. doi: 10.1084/jem.146.2.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorley-Lawson D. A., Nadler L. M., Bhan A. K., Schooley R. T. BLAST-2 [EBVCS], an early cell surface marker of human B cell activation, is superinduced by Epstein Barr virus. J Immunol. 1985 May;134(5):3007–3012. [PubMed] [Google Scholar]
- Thorley-Lawson D. A., Schooley R. T., Bhan A. K., Nadler L. M. Epstein-Barr virus superinduces a new human B cell differentiation antigen (B-LAST 1) expressed on transformed lymphoblasts. Cell. 1982 Sep;30(2):415–425. doi: 10.1016/0092-8674(82)90239-2. [DOI] [PubMed] [Google Scholar]
- Thorley-Lawson D. A., Strominger J. L. Reversible inhibition by phosphonoacetic acid of human B lymphocyte transformation by Epstein-Barr virus. Virology. 1978 May 15;86(2):423–431. doi: 10.1016/0042-6822(78)90082-x. [DOI] [PubMed] [Google Scholar]
- Wallace L. E., Rickinson A. B., Rowe M., Epstein M. A. Epstein-Barr virus-specific cytotoxic T-cell clones restricted through a single HLA antigen. Nature. 1982 Jun 3;297(5865):413–415. doi: 10.1038/297413a0. [DOI] [PubMed] [Google Scholar]
- Whitfield J. F., Rixon R. H., MacManus J. P., Balk S. D. Calcium, cyclic adenosine 3',5'-monophosphate, and the control of cell proliferation: a review. In Vitro. 1973 Jan-Feb;8(4):257–278. doi: 10.1007/BF02615905. [DOI] [PubMed] [Google Scholar]
- Yata J., Desgranges C., Nakagawa T., Favre M. C., De-The G. Lymphoblastoid transformation and kinetics of appearance of viral nuclear antigen (EBNA) in cord-blood lymphocytes infected by Epstein-Barr Virus (EBV). Int J Cancer. 1975 Mar 15;15(3):377–384. doi: 10.1002/ijc.2910150303. [DOI] [PubMed] [Google Scholar]
- de-Thé G., Geser A., Day N. E., Tukei P. M., Williams E. H., Beri D. P., Smith P. G., Dean A. G., Bronkamm G. W., Feorino P. Epidemiological evidence for causal relationship between Epstein-Barr virus and Burkitt's lymphoma from Ugandan prospective study. Nature. 1978 Aug 24;274(5673):756–761. doi: 10.1038/274756a0. [DOI] [PubMed] [Google Scholar]
- van Santen V., Cheung A., Kieff E. Epstein-Barr virus RNA VII: size and direction of transcription of virus-specified cytoplasmic RNAs in a transformed cell line. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1930–1934. doi: 10.1073/pnas.78.3.1930. [DOI] [PMC free article] [PubMed] [Google Scholar]