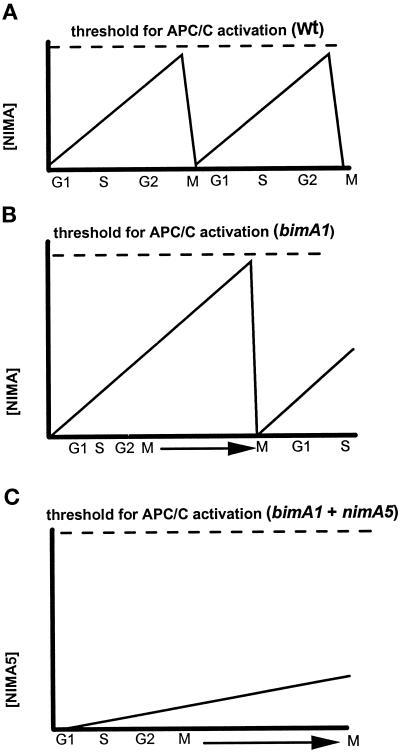
Figure 8.
Relationship between NIMA and BIMA1APC3. To help explain why the nimA5 mutation in combination with bimA1APC3 causes an effective mitotic arrest phenotype, whereas the bimA1APC3 mutation generates repeating rounds of mitotic oscillation, we propose the following. (A) Substrates of the APC/C, such as cyclin B, Polo-like kinases, and NIMA, accumulate to a threshold during mitosis, which activates the APC/C to trigger their demise and also to exit from mitosis. (B) The bimA1APC3 mutation increases the threshold of activation of the APC/C. This results in a mitotic delay during which NIMA protein and activity accumulate. When NIMA activity reaches the higher threshold level of activation, the APC/C is activated, and cells can exit mitosis. (C) In the bimA1 + nimA5 double mutant, although p34cdc2 H1 kinase activity and NIMA5 activity are increased to allow entry into mitosis, the level of NIMA5 activity stays below that required to activate the APC/C. This then causes an extended mitotic arrest, because cells cannot exit mitosis.