Abstract
Isolates of the causal ascomycete of grapevine powdery mildew, Erysiphe necator, correspond to two genetically differentiated groups (A and B) that coexist on the same host. This coexistence was analyzed by investigating temporal changes in the genetic and phenotypic structures of E. necator populations during three epidemics. Group A was present only at the start of the growing season, whereas group B was present throughout all three epidemics. Group A was less aggressive in terms of germination and infection efficiency but was more aggressive than group B in terms of the latency period, lesion diameter, and spore production. Our results are consistent with a temporal differentiation of niches, preventing recombination, and suggest an association between the disease level and the frequencies of genetic groups.
There are several well-documented examples of plant pathogen populations consisting of genetically differentiated clades in complete or partial reproductive isolation. Such a situation has been reported previously for several fungal species, including Leptosphaeria maculans (36, 39), Gaeumannomyces graminis var. tritici (5, 23), Botrytis cinerea (18), and Erysiphe syringae (35). The discovery of genetically differentiated but morphologically indistinguishable sibling species raises important questions about the coexistence of these species. According to competition theory, two species occupying the same ecological niche cannot coexist indefinitely (20). This theory has led to the suggestion that sibling species may coexist through niche partitioning, despite their high degrees of phenotypic similarity (6). For plant pathogen subgroups coexisting on the same host (i.e., the same resource), ecological differences leading to niche partitioning may result from the spatial and/or temporal separation of these subgroups (17).
Grapevine powdery mildew, which is caused by the biotrophic ascomycete Erysiphe necator (syn. Uncinula necator), provides a good model system for investigating the effects of the genetic structures of plant pathogens on plant disease epidemics. E. necator populations are structured into two genetically differentiated groups (A and B) that have been identified previously in a number of grapevine-growing countries, including France (11), Australia (14), Italy (26), and Spain (29). Délye et al. (11) suggested that group A isolates survive the winter as a resting mycelium within dormant buds, from which growth is reinitiated after budbreak, leading to the colonization of young flag shoots (30, 34); in contrast, group B isolates originate from ascospores released from overwintering cleistothecia (19). An association between flag shoot symptoms and infection with group A isolates has also been reported in other studies (1, 10, 26). However, the association between the genetic group and the overwintering strategy has been challenged by the findings in recent studies reporting that flag shoot symptoms may be caused by both groups (8, 28, 29, 32, 41).
Polymorphism in relation to E. necator aggressiveness has been poorly investigated. Recent comparisons of the degrees of aggressiveness of different E. necator genetic groups have yielded partly conflicting results: Péros et al. (31) reported that the index of infection (i.e., the proportion of sporulating lesions generated from local inoculation) was lower and that the lesion diameter was smaller for group A isolates than for group B isolates, whereas Willocquet et al. (41) found that group B isolates were more aggressive than group A isolates for certain traits (the germination ratio and infection efficiency) and that the opposite was true for other traits (the latency period, lesion diameter, and intensity of sporulation).
The frequencies of E. necator genetic groups vary considerably among vineyards, and the two groups may coexist in the same vineyard (1, 4, 7, 8, 28, 41). This finding suggests that the maintenance of a highly differentiated genetic structure in E. necator populations may result from the temporal isolation of genetic groups. This temporal pattern may result from differences in aggressiveness (i.e., quantitative pathogenicity) between isolates from different genetic groups. Therefore, this study addressed three main issues: (i) the relative proportions of A and B isolates during the course of powdery mildew epidemics, (ii) possible differences in aggressiveness traits between genetic groups of E. necator isolates, and (iii) possible changes in the aggressiveness of E. necator populations during epidemics.
MATERIALS AND METHODS
Vineyard sampling.
A preliminary sampling was conducted on 27 April 2007 in 32 commercial vineyards growing grapevines of the Carignan cultivar in the Languedoc-Roussillon region of southern France. Depending on the disease level, 6 to 37 infected leaves per vineyard were collected. Molecular characterization of the isolates present on the sampled leaves (identifying them as group A or B) was carried out immediately to facilitate field selection, and growers were asked to leave an untreated plot in each vineyard of interest.
Three of the 32 vineyards containing both E. necator genetic groups were selected for subsequent investigation. These vineyards were located at Peyriac-Minervois (PYM; 43°17′49.1″N, 2°33′27.9″E), Villarzel-Cabardès (VLZ; 43°16′46.5″N, 2°27′53.7″E), and Rieux-Minervois (RXM; 43°16′28.7″N, 2°36′30.8″E). Untreated plots of seven rows of 54 vines, six rows of 47 vines, and five rows of 49 vines at PYM, VLZ, and RXM, respectively, were used for sampling and disease assessment.
Isolate collection.
Sampling was carried out in each of the three vineyards on five dates throughout the 2007 cropping season (22 May, 20 June, 10 July, 31 July, and 27 August). Forty to 55 infected leaves with single lesions were randomly collected from each field on each date, leading to a total of 720 samples. Leaves were kept abaxial face uppermost on moist filter paper in petri dishes, and the dishes were incubated at 22°C (85 microeinstein m−2 s−1, with a 12-h/12-h photoperiod).
Assessment of disease severity.
On each sampling date, the degrees of disease severity on the leaves and clusters of two canes from each of 24 vines randomly distributed throughout the untreated parts of each of the three vineyards were assessed. For each cane, the diseased leaf or cluster area was estimated visually and expressed as a percentage of the total leaf or cluster area. The mean stages of development of the same 24 vines on each sampling date were also assessed according to the BBCH (biologische Bundesanstalt, Bundessortenamt and chemical industry) scale (21).
Measurements of components of aggressiveness.
Two days after collection, the spores from each diseased leaf sample were used as the inoculum for direct point inoculation (31). In this process, an inoculating needle was used to apply 20 to 60 conidia at the center of the upper surface of an additional leaf of grapevine cultivar Carignan. These leaves were collected from 2-month-old cuttings of vines grown in greenhouses and were disinfected in a bath of calcium hypochlorite (50 g/liter) for 10 min, rinsed in sterilized water, and dried between layers of sterile filter paper. Inoculated leaves were placed onto agar medium (20 g/liter) supplemented with benzimidazole (30 mg/liter) in petri dishes, and the dishes were incubated at 22°C.
Isolates from PYM and VLZ were characterized on the basis of a set of aggressiveness traits, the aggressiveness components (the latency period, lesion diameter, number of spores per lesion, germination ratio, and infection efficiency). No data were available for isolates from RXM, because inoculations with those isolates were unsuccessful. The reasons for this failure to isolate the fungus after inoculation were unclear but may have included the effects of antagonists (see, e.g., references 15 and 27).
The latency period was determined by observing the appearance of conidia daily under a dissecting microscope. Lesion diameters were measured 11 days after inoculation by placing a transparent sheet of paper marked with 3- to 15-mm-diameter circles over each lesion. Lesion samples were cut into two equal parts 12 days after inoculation. One half of the lesion sample was washed in a vial containing 10 ml of Isoton II (saline buffer) supplemented with a drop of dispersant type IIIA, and the number of spores produced per lesion was determined with a Multisizer 3 Coulter Counter (Beckman Coulter France, Villepinte). The remaining half of the lesion sample was used to inoculate a new leaf of cultivar Carignan, by gentle rubbing against the upper side of the leaf, to determine the germination ratio and infection efficiency, as defined by Zadoks and Schein (42). Three days after inoculation, germination ratios and infection efficiencies were estimated by using the adhesive-tape method (40) and assessing 50 spores per leaf.
Molecular characterization.
The β-tubulin gene of E. necator (tub2; GenBank accession number AY074934), with a T/C single nucleotide polymorphism differentiating between groups A and B (1), was specifically amplified. The single nucleotide polymorphism creates a recognition site for the restriction endonuclease AccI, making it possible to characterize isolates as group A or B by cleaved amplified polymorphic sequence analysis (3). DNA extraction, PCR, and cleavage reactions were carried out as described previously (28).
Data analyses.
Mixes of A and B isolates were detected only at VLZ, on three sampling dates. Two separate analyses were thus performed. A first one with VLZ samples allowed the testing of the effects of the genetic group and the sampling date on aggressiveness. A second one, based on results obtained for genotype B samples from all sites and sampling dates, allowed the assessment of the effect of the sampling date on the aggressiveness of genotype B.
The degrees of aggressiveness of group A and B isolates collected from VLZ on the first three sampling dates were compared by assessing the effects of the sampling date, the genetic group, and the combination of these two factors on each aggressiveness component by using mixed models and a split-plot design, with the sampling date as the main unit fixed effect, the genetic group as a subunit fixed effect, and the isolate as a random effect. The analysis was carried out with the MIXED procedure of the SAS software (SAS Institute Inc., Cary, NC).
Changes in the aggressiveness of genetic group B samples from PYM and VLZ during the course of the epidemics were analyzed by assessing the effects of the sampling date on each aggressiveness component by using mixed models with the sampling date as a fixed effect and the isolate as a random effect. Whenever significant effects were detected, means were compared using the Student-Newman-Keuls (SNK) test (P = 0.05).
RESULTS
Epidemic development.
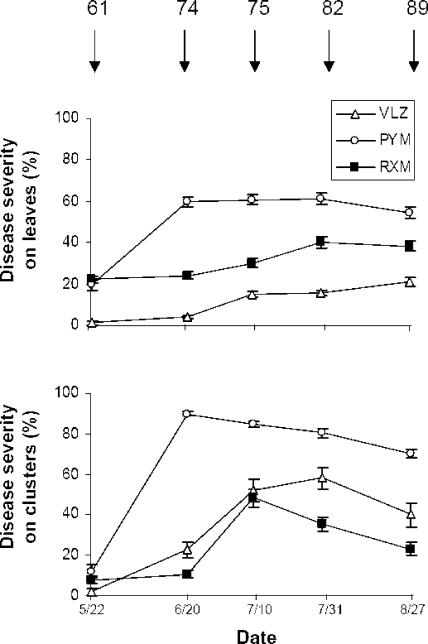
The powdery mildew epidemic took hold more rapidly and was associated with a higher final degree of severity at PYM than at RXM and VLZ (Fig. 1). The maximum severity on leaves and clusters was reached at BBCH development stage 74 (74BBCH) of the vines (characterized by the presence of small berries) only at PYM, whereas it was reached between 75BBCH and 89BBCH (characterized by the presence of pea-sized berries and berries ripe for harvest) in the other two vineyards.
FIG. 1.
Degrees of powdery mildew severity (means ± standard errors of the means [SEM]) on leaves and on clusters during the course of the 2007 growing season in three vineyards (VLZ, PYM, and RXM) of southern France. The diseased areas of leaves and clusters are expressed as percentages of the total leaf and cluster areas. Numbers at the top of the figure indicate the developmental stages according to the BBCH scale (21). Dates are given as month/day.
Changes in genetic group frequencies.
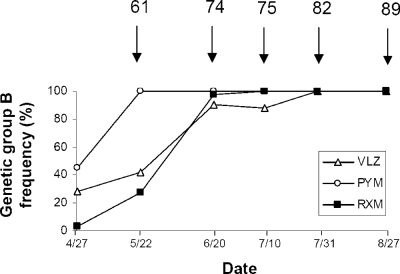
The frequencies of genetic group B isolates were 3.1% at RXM, 28.6% at VLZ, and 45% at PYM at the start of the epidemics (the end of April). The frequencies of genetic group B isolates in all three populations rapidly increased to 100% (Fig. 2). The increase in the frequency of group B isolates from PYM was more rapid than those of group B isolates from RXM and VLZ. E. necator samples from PYM contained exclusively group B isolates from the time of the first sampling (22 May; vines were at 61BBCH, i.e., the beginning of flowering), those from RXM did so from the time of the third sampling (10 July; vines were at 75BBCH), and those from VLZ contained exclusively group B isolates from the time of the fourth sampling (31 July; vines were at 82BBCH). However, from the time of the second sampling (20 June; vines were at 74BBCH), more than 90% of isolates in all populations belonged to genetic group B.
FIG. 2.
Changes during the course of the 2007 growing season in the frequencies of genetic group B isolates in E. necator populations collected from three vineyards (VLZ, PYM, and RXM) in southern France. Numbers at the top of the figure indicate the developmental stages according to the BBCH scale (21). Dates are given as month/day.
Aggressiveness.
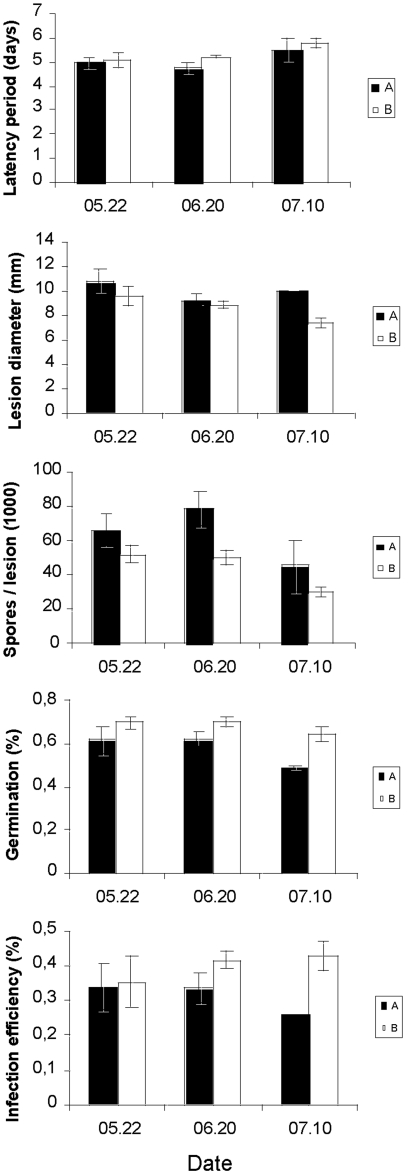
The statistical analysis of aggressiveness components for the VLZ samples revealed no significant effect of the sampling date and no significant relationship between the sampling date and the genetic group (Table 1 and Fig. 3). Comparisons of the degrees of aggressiveness of group A and group B isolates showed that genetic group A was more aggressive than genetic group B in terms of the latency period, the lesion diameter, and the number of spores produced per lesion and that group A was less aggressive than group B in terms of the germination ratio and the infection efficiency. The effect of the genetic group was significant for the number of spores produced per lesion and the germination ratio. For the latency period and the lesion diameter, the probabilities associated with F tests (P = 0.060 and 0.067, respectively) were close to the level of significance.
TABLE 1.
Results from mixed-model analyses assessing the effects of the sampling date, the genetic group, and the corresponding two-way interaction of these factors on the various aggressiveness components for E. necator isolates obtained from VLZ on the first three sampling datesa
| Aggressiveness component | Factor | Num DF | Den DF | F value | Pr > F |
|---|---|---|---|---|---|
| Latency period | Sampling date | 2 | 17 | 2.19 | 0.1424 |
| Genetic group | 1 | 12 | 4.30 | 0.0604 | |
| Date-genetic group interaction | 2 | 12 | 0.81 | 0.4658 | |
| Lesion diam | Sampling date | 2 | 17 | 2.33 | 0.1280 |
| Genetic group | 1 | 11 | 4.14 | 0.0666 | |
| Date-genetic group interaction | 2 | 11 | 0.53 | 0.6035 | |
| No. of spores/lesion | Sampling date | 2 | 17 | 3.49 | 0.0536 |
| Genetic group | 1 | 12 | 7.09 | 0.0221* | |
| Date-genetic group interaction | 2 | 12 | 0.63 | 0.5517 | |
| Germination ratio | Sampling date | 2 | 17 | 1.46 | 0.2601 |
| Genetic group | 1 | 7 | 6.31 | 0.0403* | |
| Date-genetic group interaction | 2 | 7 | 0.16 | 0.8537 | |
| Infection efficiency | Sampling date | 2 | 17 | 0.09 | 0.9103 |
| Genetic group | 1 | 7 | 2.50 | 0.1579 | |
| Date-genetic group interaction | 2 | 7 | 0.49 | 0.6322 |
Num DF, Den DF, and Pr > F are statistical terms used in SAS software indicating numerator degrees of freedom, denominator degrees of freedom, and the corresponding P value, respectively. Statistically significant effects are indicated by asterisks (P < 0.05).
FIG. 3.
Aggressiveness component values (means ± SEM) as a function of the genetic group (A or B) and sampling date for E. necator isolates collected from one French vineyard (VLZ) during the 2007 powdery mildew epidemic. Dates are given as month/day.
The degrees of aggressiveness of group B isolates from PYM did not differ significantly between the first and last sampling dates, irrespective of the aggressiveness component considered (Table 2). However, with the exception of the germination ratio, aggressiveness traits did not remain entirely stable throughout the epidemics. Isolates collected on the third sampling date showed the longest latency periods, generated lesions with the smallest diameters, produced the smallest number of spores per lesion, and, conversely, displayed the highest infection efficiencies. The same change in aggressiveness was observed for the genetic group B isolates from VLZ, but the temporal pattern of variation in aggressiveness was less clear. The least aggressive isolates (in terms of the latency period, lesion diameter, and number of spores produced per lesion) came from samples obtained on the fourth sampling date, and the most aggressive isolates (in terms of the infection efficiency) came from samples collected on the fifth sampling date.
TABLE 2.
Mean aggressiveness component values as a function of the sampling date for E. necator B isolates collected from two vineyards (PYM and VLZ) in southern Francea
| Sampling date | Mean ± SEM (SNK test group) for PYM isolates for:
|
Mean ± SEM (SNK test group) for VLZ isolates for:
|
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Latency period (days) | Lesion diam (mm) | No. of spores/lesion (1,000) | Germination ratio (%) | Infection efficiency (%) | Latency period (days) | Lesion diam (mm) | No. of spores/lesion (1,000) | Germination ratio (%) | Infection efficiency (%) | |
| 22 May | 5.72 ± 0.15 (B) | 7.81 ± 0.45 (A) | 31.55 ± 3.17 (A) | 0.68 ± 0.02 (A) | 0.39 ± 0.02 (B) | 5.10 ± 0.28 (C) | 9.67 ± 0.80 (A) | 51.83 ± 4.69 (A) | 0.70 ± 0.03 (A) | 0.3 ± 0.07 (A) |
| 20 June | 5.67 ± 0.19 (B) | 6.48 ± 0.31 (B) | 32.67 ± 2.97 (A) | 0.73 ± 0.02 (A) | 0.50 ± 0.03 (A) | 5.22 ± 0.10 (C) | 8.81 ± 0.30 (A) | 49.56 ± 3.89 (A) | 0.71 ± 0.02 (A) | 0.41 ± 0.03 (A) |
| 10 July | 6.88 ± 0.44 (A) | 5.63 ± 0.50 (B) | 16.22 ± 2.95 (B) | 0.70 ± 0.03 (A) | 0.50 ± 0.03 (A) | 5.78 ± 0.22 (B) | 7.33 ± 0.44 (B) | 30.23 ± 3.32 (B) | 0.65 ± 0.04 (A) | 0.43 ± 0.04 (A) |
| 31 July | 6.00 ± 0.26 (AB) | 6.50 ± 0.58 (AB) | 17.72 ± 2.46 (B) | 0.69 ± 0.04 (A) | 0.48 ± 0.04 (AB) | 6.57 ± 0.30 (A) | 5.43 ± 0.30 (C) | 14.72 ± 1.83 (B) | 0.72 ± 0.06 (A) | 0.51 ± 0.08 (A) |
| 27 August | 6.33 ± 0.21 (AB) | 6.42 ± 0.66 (AB) | 27.19 ± 3.46 (AB) | 0.75 ± 0.04 (A) | 0.48 ± 0.06 (AB) | 6.00 ± 0.00 (AB) | 5.63 ± 0.63 (BC) | 19.50 ± 1.07 (B) | 0.79 ± 0.03 (A) | 0.57 ± 0.06 (A) |
| F | F(4.52) = 3.05 | F(4.51) = 2.79 | F(4.51) = 3.35 | F(4.51) = 1.07 | F(4.51) = 3.06 | F(4.26) = 8.33 | F(4.25) = 9.64 | F(4.25) = 7.70 | F(4.22) = 0.95 | F(4.22) = 1.61 |
| P | 0.0247* | 0.0361* | 0.0165* | 0.3799 | 0.0247* | 0.0002*** | <0.0001*** | 0.0003*** | 0.4534 | 0.2083 |
Letters in parentheses indicate the homogeneous groups identified by the SNK test at the 5% threshold. Means followed by the same letter are not statistically different. F and P values obtained in mixed-model analyses are listed; statistically significant sampling date effects are indicated by asterisks (*, P < 0.05, and ***, P < 0.001).
DISCUSSION
Our results support the hypothesis of temporal niche differentiation to account for the co-occurrence of two genetically differentiated groups of E. necator ascomycetes. They are consistent with the findings in previous reports indicating the predominance of group A and B isolates at the beginnings and ends of epidemics, respectively (10, 12, 26). Group A isolates were present in samples acquired at the beginning of the growing season but disappeared from samples collected over the course of the epidemics, whereas group B isolates were present throughout the epidemics. Cleistothecia are produced by sexual reproduction at the end of the growing season (19). These observations, therefore, suggest that the sexual reproduction of E. necator in vineyards involves only group B isolates. It is noteworthy that the lack of sexual recombination in group A populations has been suggested previously by the findings of studies showing low levels of genotypic diversity and the presence of only one mating type in group A (11, 32). The temporal isolation of genetic groups shown in our study thus prevents recombination between group A and B isolates, accounting for the maintenance of the highly differentiated genetic structures in E. necator populations, despite demonstrations of interfertility between these groups in controlled crossing experiments (26, 37).
Group B isolates may overwinter either within buds (8, 29, 32, 41) or within cleistothecia formed by sexual reproduction, whereas group A isolates probably survive the winter exclusively within buds, which become infected at the beginning of the growing season (30). From an evolutionary point of view, this difference in survival strategy between the two genetic groups should confer an advantage on group B isolates, as these isolates benefit from both clonal and sexual reproduction. The long-term maintenance of group A isolates raises questions about the competition between E. necator genetic groups for bud infection early in the season.
A comparison of life history traits determining aggressiveness between the two genetic groups showed that group A was less aggressive than group B in terms of the germination ratio and infection efficiency and that group A was more aggressive than group B in terms of the latency period, lesion diameter, and number of spores per lesion. The findings of this study show that E. necator aggressiveness traits can be classified into two independent groups: one including monocyclic variables associated with infection (the germination ratio and infection efficiency) and the second including variables associated with epidemiological processes following infection (the latency period, spore production, and lesion size). In powdery mildew epidemics, two sympatric genetic groups may adopt different ecological strategies to colonize and use the same resource. Genetic group A isolates produce large numbers of conidia with a low infection efficiency, whereas genetic group B isolates produce smaller numbers of conidia with a higher infection efficiency. From an ecological point of view, group A isolates may be considered to have a life strategy of the [r] type, whereas group B isolates have a life strategy of the [K] type (25, 33). Invaders of vacant ecological niches tend to be [r] type strategists, whereas competitors in occupied niches tend to be [K] type strategists (24). This pattern is consistent with the observations that group A isolates were active mostly at the beginning of the growing season, subsequently disappearing, and that the frequency of group B isolates increased over time.
Our results show that aggressiveness within the two genetic groups did not increase during the course of the epidemics. This result contrasts with those obtained previously for other fungal plant pathogens. Villareal and Lannou (38) and Andrivon et al. (2) showed that directional selection for increased aggressiveness takes place in Erysiphe graminis and Phytophthora infestans populations, respectively, during the epidemic stage. Some fungal plant pathogens alternate between highly aggressive life stages and life stages in which highly aggressive strains are selected against, whereas the aggressiveness of other fungal plant pathogens, such as Mycosphaerella graminicola (9) and E. necator, remains stable.
The findings of this study demonstrate the temporal isolation of genetic groups of E. necator populations but provide no explanation for the predominance of group A at the beginning of the growing season only. However, our results did show that the disappearance of group A isolates during epidemics was not associated with an effect of fungicide, as all samples were collected from untreated plots. The data for aggressiveness components showed that numerical differences between the genetic groups were small and potentially insufficient to account for the disappearance of group A isolates during epidemics. The mechanisms underlying the apparent disappearance of group A isolates may involve the effects of temperature and/or the ontogenic resistance of leaves (13, 16).
The survival strategies of group A and B isolates seem to have major consequences for subsequent disease development and severity. The degree of disease severity, both on leaves and clusters, in the vineyard in which the frequency of group B isolates increased most rapidly was higher than those in the other two vineyards, suggesting an association between the disease level and the frequencies of genetic groups at the beginning of the growing season. A similar association between the genotype frequency and the disease level was found previously for wheat take-all (22). The results presented here are based on samples from a very small number of vineyards, but Montarry et al. (28) previously observed a strong association between the levels of disease severity at the end of the growing season and the initial compositions of E. necator populations in 13 commercial vineyards. Damage was found to be greater for epidemics initiated by group B isolates. Further studies are required to confirm these findings and may lead, in the short term, to the development of new diagnostic methods based on the early assessment of E. necator genetic groups, which would be useful for integrated crop management in vineyards.
Acknowledgments
This work was carried out as part of a research project (no. 20061202001) supported by the Région Aquitaine.
We thank C. Cabot, J. Gastou, and G. Lalaque for allowing us to use their untreated plots. We also thank F. Boyer (CA-11) for the assessment of epidemic development. We thank S. Savary, C. Robin, and D. Andrivon for very valuable comments on a draft version of this paper.
Footnotes
Published ahead of print on 22 August 2008.
REFERENCES
- 1.Amrani, L., and M. F. Corio-Costet. 2006. A single nucleotide polymorphism in the beta-tubulin gene distinguishing two genotypes of Erysiphe necator expressing different symptoms on grapevine. Plant Pathol. 55:505-512. [Google Scholar]
- 2.Andrivon, D., F. Pilet, J. Montarry, M. Hafidi, R. Corbière, E. H. Achbani, R. Pellé, and D. Ellissèche. 2007. Adaptation of Phytophthora infestans to partial resistance in potato: evidence from French and Moroccan populations. Phytopathology 97:338-343. [DOI] [PubMed] [Google Scholar]
- 3.Baudoin, A., G. Olaya, F. Delmotte, J. F. Colcol, and H. Sierotzki. 11 February 2008, posting date. QoI resistance of Plasmopara viticola and Erysiphe necator in the mid-Atlantic United States. Plant Health Prog. doi: 10.1094/PHP-2008-0211-02-RS. [DOI]
- 4.Bouscaut, J., and M. F. Corio-Costet. 2007. Detection of a specific transposon in Erysiphe necator from grapevines in France. J. Phytopathol. 155:81-383. [Google Scholar]
- 5.Bryan, G. T., M. J. Daniels, and A. E. Osbourn. 1995. Comparison of fungi within the Gaeumannomyces-Phialophora complex by analysis of ribosomal DNA sequences. Appl. Environ. Microbiol. 61:681-689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chesson, P. 2000. Mechanisms of maintenance of species diversity. Annu. Rev. Ecol. Syst. 31:343-366. [Google Scholar]
- 7.Cortesi, P., M. P. Ottaviani, and M. G. Milgroom. 2004. Spatial and genetic analysis of flag shoot subpopulation of Erysiphe necator in Italy. Phytopathology 94:544-550. [DOI] [PubMed] [Google Scholar]
- 8.Cortesi, P., A. Mazzoleni, C. Pizzatti, and M. G. Milgroom. 2005. Genetic similarity of flag shoot and ascospore subpopulations of Erysiphe necator in Italy. Appl. Environ. Microbiol. 71:7788-7791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cowger, C., and C. C. Mundt. 2002. Aggressiveness of Mycosphaerella graminicola isolates from susceptible and partially resistant wheat cultivars. Phytopathology 92:624-630. [DOI] [PubMed] [Google Scholar]
- 10.Délye, C., and M. F. Corio-Costet. 1998. Origin of primary infections of grape by Uncinula necator: RAPD analysis discriminates two biotypes. Mycol. Res. 102:283-288. [Google Scholar]
- 11.Délye, C., F. Laigret, and M. F. Corio-Costet. 1997. RAPD analysis provides insight into the biology and epidemiology of Uncinula necator. Phytopathology 87:670-677. [DOI] [PubMed] [Google Scholar]
- 12.Délye, C., V. Ronchi, F. Laigret, and M. F. Corio-Costet. 1999. Nested allele-specific PCR primers distinguish genetic groups of Uncinula necator. Appl. Environ. Microbiol. 65:3950-3954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Doster, M. A., and W. C. Schnathorst. 1985. Effects of leaf maturity and cultivar resistance on development of the powdery mildew fungus on grapevines. Phytopathology 75:318-321. [Google Scholar]
- 14.Evans, K. J., D. L. Whisson, B. E. Stummer, and E. S. Scott. 1997. DNA markers identify variation in Australian populations of Uncinula necator. Mycol. Res. 101:923-932. [Google Scholar]
- 15.Falk, S. P., D. M. Gadoury, R. C. Pearson, and R. C. Seem. 1995. Partial control of grape powdery mildew by the mycoparasite Ampelomyces quisqualis. Plant Dis. 79:483-490. [Google Scholar]
- 16.Ficke, A., D. M. Gadoury, and R. C. Seem. 2002. Ontogenic resistance and plant disease management: a case study of grape powdery mildew. Phytopathology 92:671-675. [DOI] [PubMed] [Google Scholar]
- 17.Fitt, B. D. L., Y. J. Huang, F. van den Bosch, and J. S. West. 2006. Coexistence of related pathogen species on arable crops in space and time. Annu. Rev. Phytopathol. 44:163-182. [DOI] [PubMed] [Google Scholar]
- 18.Fournier, E., T. Giraud, C. Albertini, and Y. Brygoo. 2005. Partition of the Botrytis cinerea complex in France using multiple gene genealogies. Mycologia 97:1251-1267. [DOI] [PubMed] [Google Scholar]
- 19.Gadoury, D. M., and R. C. Pearson. 1988. Initiation, development, dispersal and survival of cleistothecia of Uncinula necator in New York vineyards. Phytopathology 78:1413-1421. [Google Scholar]
- 20.Gausse, G. F. 1934. The struggle for existence. Williams and Wilkins Publishers, Baltimore, MD.
- 21.Lancashire, P. D., H. Bleiholder, T. van den Boom, P. Langeluddeke, and R. Stauss. 1991. A uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol. 119:561-601. [Google Scholar]
- 22.Lebreton, L., M. Gosme, P. Lucas, A. Y. Guillerm-Erckelboudt, and A. Sarniguet. 2007. Linear relationship between Gaeumannomyces graminis var. tritici (Ggt) genotypic frequencies and disease severity on wheat roots in the field. Environ. Microbiol. 9:492-499. [DOI] [PubMed] [Google Scholar]
- 23.Lebreton, L., P. Lucas, F. Dugas, A.-Y. Guillerm, A. Schoeny, and A. Sarniguet. 2004. Changes in population structure of the soilborne fungus Gaeumannomyces graminis var. tritici during continuous wheat cropping. Environ. Microbiol. 6:1174-1185. [DOI] [PubMed] [Google Scholar]
- 24.Lodge, D. M. 1993. Biological invasions: lessons for ecology. Trends Ecol. Evol. 8:133-137. [DOI] [PubMed] [Google Scholar]
- 25.MacArthur, R. H., and E. O. Wilson. 1967. The theory of island biogeography. Princeton University Press, Princeton, NJ. [Reprint, Princeton University Press, Princeton, NJ, 2001.]
- 26.Miazzi, M., H. Hajjeh, and F. Faretra. 2003. Observations on the population biology of the grape powdery mildew fungus Uncinula necator. J. Plant Pathol. 85:123-129. [Google Scholar]
- 27.Mmbaga, M. T., R. J. Sauve, and F. A. Mrema. 2008. Identification of microorganisms for biological control of powdery mildew in Cornus florida. Biol. Control 44:67-72. [Google Scholar]
- 28.Montarry, J., P. Cartolaro, S. Richard-Cervera, and F. Delmotte. 8 July 2008, posting date. Spatio-temporal distribution of Erysiphe necator genetic groups and their relationship with disease level in vineyards. Eur. J. Plant Pathol. doi: 10.1007/s10658-008-9343-9. [DOI]
- 29.Nuñez, Y., J. Gallego, F. Ponz, and R. Raposo. 2006. Analysis of population structure of Erysiphe necator using AFLP markers. Plant Pathol. 55:650-656. [Google Scholar]
- 30.Pearson, R. C., and W. Gärtel. 1985. Occurrence of hyphae of Uncinula necator in buds of grapevine. Plant Dis. 69:149-151. [Google Scholar]
- 31.Péros, J. P., T. H. Nguyen, C. Troulet, C. Michel-Romiti, and J. L. Notteghem. 2006. Assessment of powdery mildew resistance of grape and Erysiphe necator pathogenicity using a laboratory assay. Vitis 45:29-36. [Google Scholar]
- 32.Péros, J. P., C. Troulet, M. Guerriero, C. Michel-Romiti, and J. L. Notteghem. 2005. Genetic variation and population structure of the grape powdery mildew fungus, Erysiphe necator, in southern France. Eur. J. Plant Pathol. 113:407-416. [Google Scholar]
- 33.Pianka, E. R. 1970. On r and K selection. Am. Nat. 104:592-597. [Google Scholar]
- 34.Sall, M. A., and J. Wrysinski. 1982. Perennation of powdery mildew in buds of grapevines. Plant Dis. 66:678-679. [Google Scholar]
- 35.Seko, Y., A. Bolay, L. Kiss, V. Heluta, B. Grigaliunaite, and S. Takamatsu. 2008. Molecular evidence in support of recent migration of a powdery mildew fungus on Syringa spp. into Europe from East Asia. Plant Pathol. 57:243-250. [Google Scholar]
- 36.Shoemaker, R. A., and H. Brun. 2001. The teleomorph of the weakly aggressive segregate of Leptosphaeria maculans. Can. J. Bot. 79:412-419. [Google Scholar]
- 37.Stummer, B. E., and E. S. Scott. 2003. Detection of novel genotypes in progeny from a controlled cross between isolates of Uncinula necator belonging to distinct phonetic groups. Aust. Plant Pathol. 32:213-218. [Google Scholar]
- 38.Villareal, L. M. M. A., and C. Lannou. 2000. Selection for increased spore efficacy by host genetic background in a wheat powdery mildew population. Phytopathology 90:1300-1306. [DOI] [PubMed] [Google Scholar]
- 39.Williams, R. H., and B. D. L. Fitt. 1999. Differentiating A and B groups of Leptosphaeria maculans, causal agent of stem canker (blackleg) of oilseed rape. Plant Pathol. 48:161-175. [Google Scholar]
- 40.Willocquet, L., D. Colombet, M. Rougier, J. Fargues, and M. Clerjeau. 1996. Effect of radiation, especially UV-B, on spore germination and mycelial growth of the grape powdery mildew. Eur. J. Plant Pathol. 102:441-449. [Google Scholar]
- 41.Willocquet, L., P. Cartolaro, J. Jolivet, S. Richard-Cervera, and F. Delmotte. 2007. Relationships between genetic group, symptom type, and epidemiological features in Erysiphe necator, the causal agent of grape powdery mildew. Phytopathology 97(Suppl.):S123. [Google Scholar]
- 42.Zadoks, J. C., and R. D. Schein. 1979. Epidemiology and plant disease management. Oxford University Press, New York, NY.