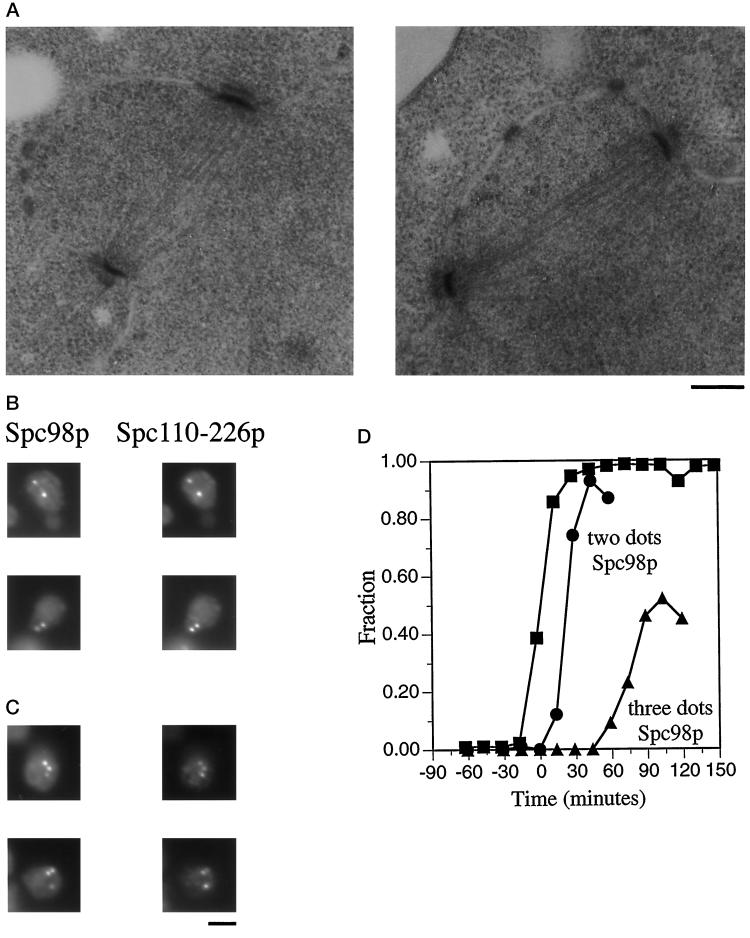
Figure 6.
spc110–226 forms normal spindles early, but spindle integrity is compromised later. A population of synchronous spc110–226 cells was obtained from strain HSY83 by centrifugal elutriation and shifted to 37°C. At regular intervals samples were removed and processed for either electron microscopy or immunofluorescence. t = 0 min is the midpoint of S phase. (A) Electron micrographs depicting short complete spindles at t = 12 min. Bar, 0.2 μm. (B) Spc98p staining and Spc110–226p staining of spc110–226 mutant cells reveal two dots of colocalized staining representing the poles of a short complete spindle 43 min after the midpoint of S phase. The cells were stained with affinity-purified anti-Spc110p antibodies (1:500) (Friedman et al., 1996) and mouse monoclonal anti-98 kDa antibodies (1:10; gift of J. Kilmartin). The secondary antibodies were a mixture of rhodamine-conjugated affinity-purified sheep IgG anti-mouse Ig (20 μg/ml) and fluorescein-conjugated goat anti-rabbit antibody (1:1000; Boehringer Mannheim Corp.). Bar, 2 μm. (C) Spc98p staining and Spc110–226p staining of spc110–226 mutant cells reveal three dots of colocalized staining and thus the presence of an aberrant structure 103 min after the midpoint of S phase in the synchronous shift experiment. Samples were processed for immunofluorescence as described in Figure 6B. Bar, 2 μm. (D) Timing of the appearance of three dots of Spc98p staining in the spc110–226 synchronous shift experiment. ▪, fraction of the cell population that has replicated at least one-half of its genome; •, fraction of spc110–226 cells with two dots of anti-98 kDa staining; ▴, fraction of spc110–226 cells with three or more dots of anti-98 kDa staining.