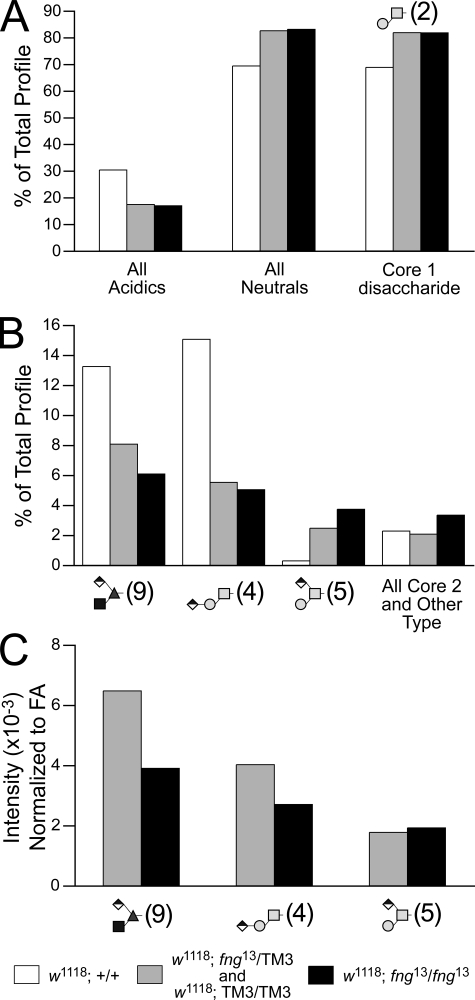
FIGURE 8.
A mutation in the fringe gene decreases the prevalence of the O-Fuc trisaccharide and the linear core 1 structure. Embryos collected from adults heterozygous for the fng13 mutation carried over a GFP balancer chromosome (TM3-GFP) were manually sorted by fluorescence (supplemental Fig. 7), yielding 884 homozygous fng13 late-stage embryos and a pool of age-matched control siblings (mixed fng13/TM3-GFP heterozygotes and TM3-GFP homozygotes). O-Linked glycans were prepared from the sorted embryos and from w1118 embryos, the genetic background in which the fng13 mutation is maintained. A, the prevalence of acidic glycans is decreased in fng mutant embryos. A corresponding increase in neutral glycans is driven almost entirely by a higher relative prevalence of the core 1 disaccharide (Structure 2). B, two of the three major glucuronylated O-linked glycans are reduced in fng mutant embryos as a percent of the total profile, including the O-Fuc trisaccharide (Structure 9), which is predicted to require Fng enzyme activity for its biosynthesis. C, normalized to total fatty acid content, the MS signal intensities for the O-Fuc trisaccharide and for the linear core 1 trisaccharide (Structure 4) decrease in proportion to the dose of the mutant fng gene. The amount of the branched, glucuronylated core 1 trisaccharide (Structure 5) is unchanged by loss of Fng function.