Figure 5.
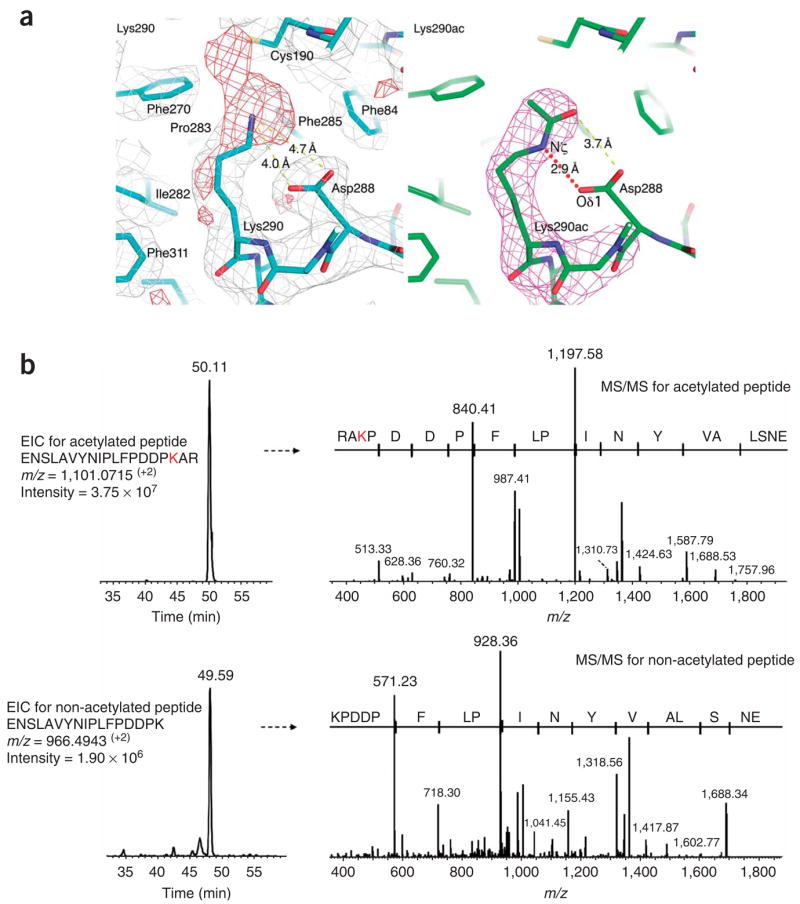
Rtt109 autoacetylation at Lys290. (a) Close-up view of acetyllysine residue 290 of Rtt109. Left: 2Fo − Fc (silver, 1σ) and Fo − Fc (red, 2.5σ) difference maps are shown after refinement of a Lys290 side chain. Right: simulated annealing omit (magenta, 2.5σ) maps are shown after refinement of an acetylated Lys290 side chain. Selected interatomic distances are highlighted in yellow dotted lines. Note the formation of a hydrogen bond mediated by the acetylated lysine side chain, between Lys290 Nζ and Asp288 Oδ1, as shown in red dotted line. (b) Extracted ion chromatogram (EIC) from LC-MS/MS analysis of the Rtt109 tryptic peptides containing acetylated Lys290 (upper left) and its corresponding MS/MS spectrum (upper right), confirming the location of the modified Lys290 residue. Lower panels show the unmodified Lys290 tryptic peptide from the same Rtt109 protein sample. Comparison of the relative intensities of the modified and nonmodified Lys290 residue further confirmed the prevalent in vivo acetylation of this residue.
