Table 2.
Chemical structures and physicochemical properties of isoleucine, methionine, and methionine sulfoxide
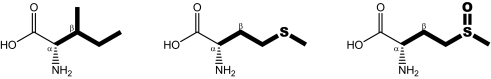 |
|||
|---|---|---|---|
| LogP | 2.38 | 1.63 | −0.50 |
| p [D] | 0.01 | 1.56 | 3.79 |
| A [Å2] | 117.7 | 122.0 | 125.0 |
| Ap [Å2] | 0.0 | 0.0 | 22.3 |
The following properties were calculated for the displayed amino acid side chains depicted in bold: LogP, octanol-water partition coefficient; p, dipole moment; A, Connolly molecular surface area; Ap, polar surface area. Transitions from isoleucine to methionine are accompanied by a loss in sterical demand in the β position, whereas the overall elongated and hydrophobic character of the side chain is largely maintained.
