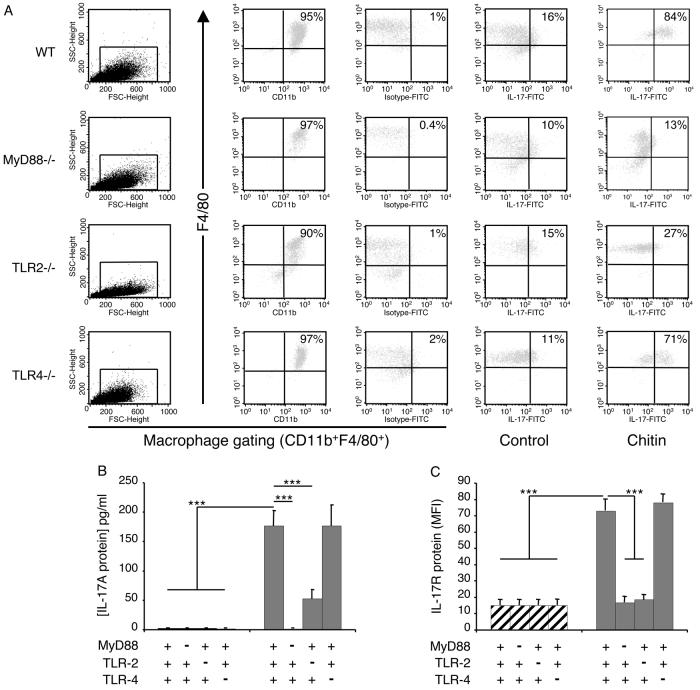
Figure 3. Mechanisms of chitin regulation of macrophage IL-17 and IL-17R.
Peritoneal macrophages were obtained from wild-type (WT), MyD88, or TLR sufficient (+/+; +) or deficient (−/−; −) mice and were incubated with chitin (40-70 μm; 100 μg/ml) or vehicle control for 8h. The effects of these interventions on intracellular IL-17 protein (A), soluble IL-17 protein (B) and IL-17R protein production (C) were evaluated. Macrophage intracellular IL-17 and IL-17R protein were assessed using FACS analysis. For intracellular IL-17 staining, cell debris was excluded by light-scatter cell gating (A; first column) and this gate was used for all subsequent evaluations. Macrophages were identified by surface marker staining using F4/80 and CD11b co-staining which revealed a macrophage purity higher than 90% for all tested conditions (A; second column). To specifically stain intracellular IL-17A, macrophages were fixed and permeabilized. Fc blocking and isotype controls were used to exclude unspecific staining (A; third column). In control (A; fourth column) and chitin (A; fifth column)-treated macrophages intracellular IL-17A staining is depicted with F4/80 co-staining. In (B), supernatant IL-17 was assessed by ELISA. In C, macrophage IL-17AR expression was assessed using FACS analysis and is expressed as the MFI of the evaluation. *** p < 0.001.