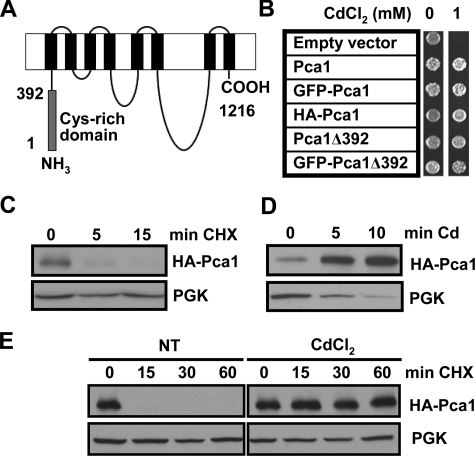
FIGURE 1.
Cadmium rapidly enhances Pca1 expression by increasing stability. A, schematic depiction of the Pca1 P1B-type ATPase. Gray and black rectangles represent the N-terminal cysteine-rich cytosolic domain encompassing the first 392 amino acids and eight predicted transmembrane domains, respectively. B, cadmium resistance in cells expressing Pca1 alleles. Pca1, N-terminal GFP-fused Pca1 (GFP-Pca1), three tandem HA epitope-tagged Pca1 (HA-Pca1), and Pca1 deleted of the N-terminal cysteine-rich domain (Pca1Δ392) with or without N-terminal GFP fusion were expressed in a BY4741 yeast strain in which endogenous Pca1 is nonfunctional (30). The cells (5 μl, A600 = 1.0) were spotted on SC solid medium containing the indicated concentration of CdCl2, and then cell growth was assessed after 2 days. C–E, HA-Pca1 expression was detected by Western blotting using anti-HA antibodies. The same blot was probed for PGK to determine equal loading. C, cycloheximide chase of yeast cells expressing HA-Pca1. Exponentially growing cells were co-cultured with cycloheximide (100 μg/ml) and then collected at the indicated time points for Western blot analysis. D, rapid up-regulation of Pca1 in response to cadmium. Cadmium (50 μm CdCl2) was added to exponentially growing yeast cultures, and then the cells were collected into ice-cold kill buffer (15 mm NaN3 PBS) at the indicated time points. E, cadmium-induced stabilization of Pca1 determined by cycloheximide chase. Yeast cells precultured for 15 min without (NT) or with cadmium (20 μm CdCl2) were collected at the indicated time points. Cycloheximide (100 μg/ml) was added to the culture medium after collection of time zero. Total protein extracts were subjected to Western blot analysis.