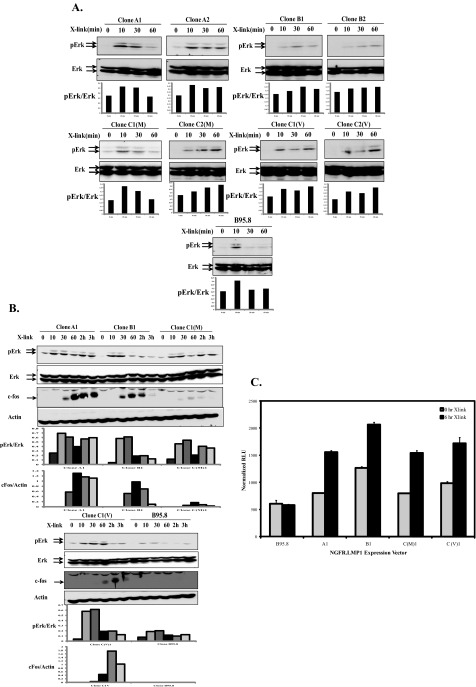
FIGURE 5.
LMP1 tumor-derived variants induce more sustained Erk activation, c-Fos, and AP-1 activity than B95.8 isoform. A, NGFR-LMP1 signaling was induced in BL41clones expressing tumor-derived LMP1 or B95.8-derived LMP1 by cross-linking with anti-NGFR and goat anti-mouse IgG for 0, 10, 30, or 60 min. Cells were lysed in phospholysis buffer, and lysates were analyzed for Erk activation by Western blot with anti-phospho-Erk (pErk) and total Erk antibodies. Arrows indicate Erk1 (top band) and Erk2 (bottom band). The corresponding densitometry graphs are shown below each blot. The blots are representative of four independent experiments. B, BL41 clones expressing NGFR-LMP1 proteins were activated by anti-NGFR and goat anti-mouse IgG cross-linking for 0, 10, 30, and 60 min and 2 and 3 h. The cells were harvested and lysed at the time point required and analyzed for Erk activation and c-Fos induction by Western blot with anti-pErk, anti-Erk, anti-c-Fos, and anti-β-actin antibodies. The corresponding densitometry graphs are shown below each blot. The blots are representative of three independent experiments. C, 293T cells were transiently transfected with firefly luciferase reporter plasmid, Renilla luciferase reporter plasmid, and NGFR-LMP1 expression plasmids. NGFR-LMP1 signaling was induced in 293T cells expressing tumor-derived LMP1 or B95.8 LMP1 by cross-linking with anti-NGFR and goat anti-mouse IgG for 0 or 6 h. Relative luciferase activity (RLU) was calculated as the ratio of firefly luciferase activity to Renilla luciferase activity as expressed as normalized RLU. Data represent the mean of triplicate samples and are representative of three independent experiments.