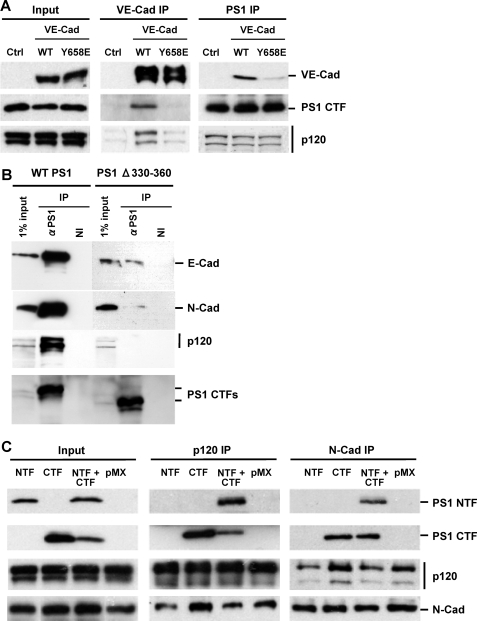
FIGURE 1.
Cadherin or PS1 mutants unable to bind p120ctn fail to form PS1-cadherin complexes. A, Chinese hamster ovary cell cultures were transfected with enhanced GFP, WT VE-cadherin-enhanced GFP, or VE-cadherin mutant Y658E-enhanced GFP, which is unable to bind p120ctn. Stable GFP-positive cell populations expressing similar levels of VE-cadherin were sorted and selected. Extracts from all cultures were immunoprecipitated with either anti-VE-cadherin (VE-cad IP)(middle panels) or anti-PS1 (PS1 IP)(right panels) antibodies, and obtained IPs were probed on WBs with antibodies against antigens indicated at the right of the figure as described (1). Input is shown in the left panels. WT VE-cadherin forms complexes with both p120ctn and PS1. In contrast, mutant VE-cadherin binds neither p120ctn nor PS1. As expected, PS1 binds p120ctn in all cultures (lower right panel). B, HEK293 cells transduced with WT PS1 and PS1Δ330–360 were grown to confluency, extracted in Nonidet P-40, and immunoprecipitated with anti-PS1 antibody (Sigma S182 polyclonal antibody) or nonimmune serum (NI). Obtained immunoprecipitated samples were probed on WBs with antibodies against E-cadherin, N-cadherin, p120ctn, and PS1/CTF indicated at the right of the figure. Deletion mutant PS1Δ330–360, which is unable to bind cadherin (24), is also unable to bind p120ctn. C, PS1–/– mouse fibroblasts were transduced with PS1/CTF, NTF, or both. Extract from each culture was prepared in Nonidet P-40 and then immunoprecipitated with either anti-p120ctn (p120ctn IP)(middle panels) or anti-N-cadherin (N-cad IP)(right panels) antibodies. Obtained IPs were probed on WBs with antibodies against antigens indicated at the right of the figure. PS1/CTF binds both p120ctn and N-cadherin, whereas PS1/NTF needs CTF to associate with p120ctn or N-cadherin.