Abstract
We examined open-field effects in rats of the cannabinoid 1 receptor (CB1R) agonist WIN55,212-2 (WIN; 3 mg/kg) and its interaction with the CB1R putative neutral antagonist AM4113 (0.3 to 3 mg/kg). Separate studies examined AM4113 alone (0.3 to 5.6 mg/kg). Unlike the CB1R antagonist rimonabant, in vitro (e.g., Sink et al., 2007) AM4113 produced no change in cAMP accumulation (neutral antagonism vis-a-vie inverse agonism). Recorded behaviors were: ambulation, rearing, circling, latency, scratching, grooming, defecation, urination and vocalization/squeaking. WIN reduced ambulation and rearing; AM4113 completely (ambulation) or partially (rearing) antagonized these behaviors. WIN alone resulted in circling and an increased latency to leave the start-area; effects blocked by AM4113. AM4113 increased scratching and grooming, effects attenuated but not abolished by WIN. AM4113 alone tended to reduce ambulation and rearing and had no effect on latency or circling. AM4113 alone increased scratching and grooming. Effects on defecation, urination and vocalization were non-significant. The open-field effects of AM4113 are similar to those reported for rimonabant in rats. Yet, unlike the inverse agonists rimonabant and AM251, the putative neutral CB1R antagonist AM4113 did not produce signs of nausea in ferrets and rats (Chambers et al., 2007; Sink et al., 2007).
Keywords: WIN55,212-2; AM4113; cannabinoid; agonist; neutral antagonist; open-field; rats
1. Introduction
The first selective cannabinoid 1 receptor (CB1R) antagonist developed was rimonabant (SR141716A; Rinaldi-Carmona et al., 1994). This discovery was followed by the synthesis of additional CB1R selective antagonists such as LY320135, SR147778, AM251, AM281, and AM1387 (Felder et al., 1998; Rinaldi-Carmona et al., 2004; Thakur et al., 2005). In vitro assays showed that higher concentrations of rimonabant decreased GTPγS binding and increased cAMP production, suggesting that this ligand has inverse agonist properties as do all of the other above CB1R antagonists by such definition (Howlett et al., 2004; Pertwee, 2005). In addition to its ability to antagonize the effects of CB1R agonists both in vitro and in vivo, rimonabant produces behavioral effects of its own. For example, increased locomotion has been described in mice (Bass et al., 2002; Compton et al., 1996; see also Cosenza et al., 2000) as well as an increased incidence of scratching (Darmani and Pandya, 2000; Janoyan et al., 2002). Likewise, increased levels of scratching and grooming have been observed for rats (Järbe et al., 2002; 2006; Navarro et al., 1997; Pavón et al., 2006; Rubino et al., 1998; 2000; Tallett et al., 2007a; Vickers et al., 2003). These characteristic rimonabant induced intrinsic effects appear centrally mediated (Rodriguez de Fonseca et al., 1998; Pavón et al., 2006), and are attenuated to varying degrees by CB1R agonists. However, it is unclear if these intrinsic effects are due to inverse agonism or if other mechanisms (e.g., change in endocannabinoid tone) are responsible for their occurrence (see overview by Pertwee, 2005). Bass et al. (2002) concluded that inverse agonism at CB1R, as determined by decreased GTPγS binding or for that matter CB1R activation alone, were not sufficient to explain the locomotor stimulatory effects of rimonabant and some of its analogs in mice.
Receptor blockers devoid of intrinsic activity per se at the cellular level, i.e., not triggering changes in cellular signaling, are classified as neutral or silent antagonists. However, it needs to be kept in mind that the distinction between neutral antagonism and inverse agonism may be system dependent as well as end-point dependent (e.g., Govaerts et al., 2004). CB1R ligands that behave as neutral antagonists in vitro have been synthesized. As recently reviewed by Pertwee (2005), O2050 (a high-affinity sulphonamide analog of Δ8-THC) behaved as a neutral antagonist in two tissue preparations (muscle and vas deferens; see also Canals and Milligan, 2008). In vivo, both rimonabant and O2050 reduced food intake but unlike 3 mg/kg rimonabant, 0.3 and 3 mg/kg O2050 produced hypoactivity in rats (Gardner and Mallet, 2006; see also Martin et al., 2002). Another neutral antagonist is NESS0327 (Ruiu et al., 2003), which has considerably higher affinities for both CB1 and CB2 receptors compared to rimonabant, but unlike rimonabant does not affect basal GTPγS binding at concentrations up to 1 µM. Additionally, in vivo NESS0327 blocked the antinociceptive effects of WIN55,212-2 (WIN), and when examined alone displayed no antinociception per se (Ruiu et al., 2003). The 1,2,4-triazole derivative LH21 (Jagerovic et al., 2004) has also been described as a neutral in vivo cannabinoid receptor antagonist (Pavón et al., 2006), but the basis for this claim appears to be reduced brain bioavailability compared to rimonabant rather than any particular differential effects on measures of inverse agonism (further elaborated in Pavón et al., 2008). Still other potential neutral CB1R antagonists are listed in Pertwee (2005) and elsewhere (Fride et al., 2007; Hurst et al., 2006). The above purported neutral antagonists have not been extensively characterized in vivo. From a medicinal point of view, the interest in developing neutral or silent antagonists is that they might exhibit fewer unwanted (side) effects compared to inverse agonists (see Greasley and Clapham 2006 for discussion). They also may serve as pharmacological tools to potentially dissociate CB1R effects related to inverse agonism or if the behavior in question is under CB1R tonic control.
The present study investigated the effects of the newly developed putative neutral CB1R antagonist AM4113, a pyrazole analog structurally related to rimonabant and AM251, in an open-field. Unlike rimonabant and AM251, AM4113 does not enhance forskolin stimulated cAMP formation in vitro, but retains the ability to reduce food intake in rodents without concomitant signs of nausea (Chambers et al., 2007; Sink et al., 2007). AM4113 was examined in vivo alone and when combined with the CB1R agonist WIN. The open-field test generates a variety of behaviors that are sensitive to drug manipulation. In addition to reducing food intake, the prototypical CB1R antagonist/inverse agonist rimonabant characteristically also increases scratching and grooming in a dose-dependent manner and decreases ambulation (horizontal activity) and perhaps to a lesser extent also rearing (vertical activity) in rats at higher doses (e.g., Järbe et al., 2002; 2006; see also McLaughlin et al., 2005 as well as Tillett et al., 2007b regarding AM251). On the other hand, CB1R agonists at moderate to high doses tend to suppress these open-field behaviors, and at high doses also induce circling (the number of times a rat turns around its vertical axis); the latter behavior is not seen either in control animals or rimonabant treated rats. In this study we examined the ability of AM4113 to antagonize WIN induced behavioral alterations and if the putative CB1R neutral antagonist AM4113 would produce a qualitatively different behavioral/open-field profile relative to rimonabant with emphasis on scratching and grooming.
2. Materials and methods
2.1. Animals
A total of 100 adult male Sprague-Dawley rats (Taconic Farms, Germantown, NY), between 2.5 to 3.5 months old upon arrival to the Northeastern quarters, were used for the open-field studies. The animals were left undisturbed for one week and thereafter the animals were handled for approximately 5 min each weekday during the two weeks prior to beginning the test phase. During the 2nd week of handling, the animals were also injected with 2 ml/kg of vehicle (see below) to accustom the animals to injection procedures. Rats were individually housed with free access to food and water under a 12 hours light/dark cycle (lights on at 7 A.M.). The Animal Care and Use Committee of Northeastern University, Boston, MA, approved all procedures. The "Principles of Animal Laboratory Care" (National Institutes of Health, 1996) was followed.
2.2. Treatments
In tests with WIN and AM4113, rats were given two i. p. injections (2 ml/kg each) on either side of the peritoneal midline 30 min prior to testing in the open-field arena; this corresponds to our previous open-field examinations with THC or WIN and rimonabant (Järbe et al., 2002; 2006). Groups of rats (n = 10) were given vehicle and vehicle (controls), WIN and vehicle or AM4113, or AM4113 and vehicle. The dose of WIN was 3 mg/kg (see Järbe et al., 2006), and the doses of AM4113 were 0.3, 1, and 3 mg/kg in the interaction studies. When examining AM4113 alone, we also included a dose of 5.6 mg/kg in addition to the three doses of AM4113 used for the interaction study with WIN.
Treatments (i. e., various combinations of drugs and dosages) were staggered such that one third of the rats in each condition completed the open-field test before initiating experiments with the second third of the subjects. This protocol was repeated prior to testing with the last third of the subjects. This precaution aimed at counterbalancing for the possible influence of length of stay in the vivarium prior to testing. Open-field sessions occurred during the lighted portion (1 to 4 PM) of the light/dark cycle during week days to minimize the influence of diurnal variations.
2.3. Open-Field test apparatus
The open-field arena is a gray painted box (60 × 60 × 45 cm) with an open top and a white floor divided into 16 squares (15 × 15 cm) and a circle (19 cm in diameter) marked in the center of the field. The floor was covered with a piece of acrylic, which was cleaned and dried between each session. A video camera was mounted 1.5 m above the floor of the open-field arena, such that the whole arena was viewable on camera. Lighting was provided by two clip-on incandescent lamps with 100-w bulbs about 2 m above the box floor. The entire apparatus and camera were centered in an otherwise empty room measuring 2.7 m × 1.1 m. A TV screen, connected to the video system (VHS VCR), was located outside of the experimental room enabling us to monitor and record the rats during open-field testing for later scoring (scoring was not blinded). Animals were transported to the experimental area about two hours before any session started. Thirty minutes after injection, the session began by placing the rat in the center circle and sessions ended after five minutes. No experimenters were in the experimental room during the 5 minutes open-field session.
2.4. Measures
The behavioral measures recorded were i) ambulation (the number of squares crossed with all four feet), ii) rearing frequency (the number of times the rat stood erect on its hind-legs), iii) latency (the time in seconds to leave the circle in the center of the field with all four feet), iv) circling (the number of times the animals turned around its vertical axis, 0.5 point given for each 180 degrees turn). We also recorded whether circling (or turning behavior) consistently was directed to the left or right and also if it shifted during a single open-field exposure; v) grooming episodes (the number of cleaning bouts), as well as vi) grooming duration (i.e., the total time in seconds spent grooming) as it has been argued that total duration time rather than just frequency of grooming is a more revealing measure (Eilam et al., 1992), though frequency is the more commonly used measure. We also kept record of vii) scratching frequency (defined according to Darmani and Pandya, 2000), i.e., "A scratching episode produced by a particular hind limb consisted of 1 or more repetitive scratches with less than 2-seconds in between. If the interval between consecutive scratches by a particular hind limb was greater than 2-seconds, the scratches were considered separate episodes. If the scratches were produced by alternative hind legs, then each scratch was considered as a separate episode". Urination and defecation (the number of urination spots and fecal boli deposited during the 5 minutes observation period) were also recorded. The presence/absence of vocalization (squeaking) was also noted when the rat was lifted up for placement into the open-field arena (“vocalization before”), as well as when the rat was lifted up for removal from the open-field arena (“vocalization after”).
2.5. Drugs
WIN55,212-2 was purchased from Sigma-Aldrich, St. Louis, MO. The novel putative neutral CB1R antagonist AM4113 (N-piperidin-1-yl-2,4-dichlorophenyl-1H-pyrazole-3-carboxamide analog) was synthesized at the Center for Drug Discovery, Northeastern University; the compound is structurally related to AM251. WIN was dissolved in a solution of propylene glycol (PG, 5%) and Tween-80 (T-80, 5%); and AM4113 in a solution consisting of DMSO (2%), PG (4%), and Emulphor-620 (4%; Rhone-Poulenc, Inc., Princeton, NJ). Shortly before being used, the solute was diluted (v/v) with normal (0.9%) saline after the solute had been sonicated for 20 minutes. All doses were administered i.p. in a volume of 2 ml/kg, 30 minutes prior to session onset.
2.6. Statistics
Completely randomized one-way analyses of variance (ANOVA) were calculated using SigmaStat (v. 3.10; Systat Software, Inc., Point Richmond, CA; www.systat.com). When ANOVA was significant, post-hoc analyses were carried out with the Holm-Sidak all pair-wise comparisons method with alpha, two-tailed set at 0.05 (i.e., for the collection of comparisons). To better meet the assumptions of homogeneity of error variances and normality of treatment-level distributions, all data were square-root transformed for statistical analysis (Kirk, 1968).
3. Results
3.1. WIN alone and in combination with AM4113
Figure 1 shows the effects of WIN (3 mg/kg) in combination with AM4113 (0, 0.3, 1, and 3 mg/kg) on ambulation (A), rearing (B), circling (C) and latency (D).
Figure 1.
The effects of WIN (3 mg/kg) in combination with AM4113 (0, 0.3, 1, and 3 mg/kg) on ambulation (A), rearing (B), circling (C) and latency to leave the center circle (D) in different groups of Sprague-Dawley rats (n = 10). WIN and AM4113 injections were given i.p. 30 minutes prior to session onset; controls received two vehicle (2 ml/kg each) injections (V+V). The data points represent the means (±SEM) during a 5-minutes observation period in an open-field arena (n = 10).
Ambulation frequency (A): *) different from controls (V+V); +) WIN+V different from WIN+AM0.3, WIN+AM1 and WIN+AM3.
Rearing frequency (B): *) different from controls (V+V); +) WIN+V different from WIN+AM0.3, WIN+AM1 and WIN+AM3.
Circling frequency (C): *) different from controls (V+V).
Latency time (D): *) different from controls (V+V); +) WIN+V different from WIN+AM0.3, WIN+AM1 and WIN+AM3.
Ambulation (A)
WIN suppressed this horizontal, exploratory behavior (see data above label WIN+V). This suppression was antagonized by AM4113 (see WIN+AM). All three doses of AM4113 brought the activity counts in the WIN (3 mg/kg) treated rats to levels comparable to that of the controls (V+V); [F (4, 45) = 13.75; p < 0.001].
Rearing (B)
WIN strongly suppressed this vertical behavior (see data above label WIN+V). The suppression was attenuated by AM4113. However, none of the three antagonist doses brought the activity counts in the WIN (3 mg/kg) treated rats to levels comparable to that of the controls (V+V). Thus, rearing levels were still significantly below control levels (graph B, label V+V); [F (4, 45) = 16.15; p < 0.001].
Circling (C)
This behavior was significantly increased by WIN (WIN+V) compared to controls (V+V), and antagonized by AM4113 (see WIN+AM) in a dose-dependent manner; [F (4, 45) = 4.26; p = 0.005].
Latency (D)
Latency to leave the middle circle of the open-field arena with all four feet was significantly increased by WIN (WIN+V) compared to the controls (V+V), and antagonized by all the three AM4113 doses examined (0.3 to 3 mg/kg; see bars in graph D above WIN+AM); [F (4, 45) = 7.53; p < 0.001].
Figure 2 shows the effects of WIN (3 mg/kg) in combination with AM4113 (0, 0.3, 1, and 3 mg/kg) on scratching (A), grooming frequency (B), and grooming duration (C).
Figure 2.
The effects of WIN (3 mg/kg) in combination with AM4113 (0, 0.3, 1, and 3 mg/kg) on scratching (A), grooming frequency (B), and grooming duration (C) in different groups of Sprague-Dawley rats (n = 10). WIN and AM4113 injections were given i.p. 30 minutes prior to session onset; controls received two vehicle (2 ml/kg each) injections (V+V). The data points represent the means (±SEM) during a 5-minutes observation period in an open-field arena (n = 10).
Scratching frequency (A): *) different from controls (V+V); +) WIN+V different from WIN+AM1 and WIN+AM3; x) WIN+AM3 different from WIN+AM0.3 and WIN+AM1.
Grooming frequency (B): x) WIN+AM3 different from WIN+V and WIN+AM0.3.
Grooming duration (C): *) different from controls (V+V); x) WIN+AM3 different from WIN+V, WIN+AM0.3 and WIN+AM1.
Scratching (A)
AM4113 (in combination with WIN) produced dose-dependent increases in the amount of scratching compared to vehicle (V+V) and WIN alone (WIN+V), significantly so with 1 and 3 mg/kg AM4113 (see bars above labels WIN+AM1 and WIN+AM3); [F (4, 45) = 12.83 p < 0.001].
Grooming frequency (B)
Grooming increased significantly with 3 mg/kg AM4113 (plus WIN 3 mg/kg) compared to WIN alone (3 mg/kg; see WIN+V); [F (4, 45) = 5.25 p = 0.001].
Grooming duration (C)
Addition of AM4113 to WIN tended to also increase the grooming duration. Thus, addition of 3 mg/kg AM4113 to 3 mg/kg WIN increased grooming duration above that seen for the controls (V+V), as well as that of 3 mg/kg of WIN alone (WIN+V); [F (4, 45) = 9.96; p < 0.001].
Defecation, Urination and Vocalization (before and after) were not statistically significant when examining WIN alone and when this CB1R agonist was combined with the putative neutral CB1R antagonist AM4113 (p > 0.05; not shown).
3.2. AM4113 alone
Figure 3 shows the effects of AM4113 (0, 0.3, 1, 3, and 5.6 mg/kg) alone on ambulation (A), rearing (B), and latency (C).
Figure 3.
The effects of AM4113 (0, 0.3, 1, 3, and 5.6 mg/kg) on ambulation (A), rearing (B), and latency to leave the center circle (C) in different groups of Sprague-Dawley rats (n = 10). AM4113 plus vehicle injections were given i.p. 30 minutes prior to session onset; controls received two vehicle (2 ml/kg each) injections (V+V). The data points represent the means (±SEM) during a 5-minutes observation period in an open-field arena (n = 10).
Ambulation frequency (A): *) different from controls (V+V).
Rearing frequency (B): *) different from controls (V+V).
Latency time (C): non-significant (p > 0.05).
Ambulation (A)
There were two instances of reduced ambulatory (horizontal) activity. Locomotion was significantly lower in the 3 (V+AM3) and 5.6 (V+AM5.6) mg/kg AM4113 treated animals compared to the controls (V+V); [F (4, 45) = 4.80; p = 0.003].
Rearing (B)
There was one statistically significant instance of reduced rearing (vertical activity). Rearing was lower in the 3, but not in the 5.6 mg/kg AM4113 treated animals compared to controls (V+V); [F (4, 45) = 2.62; p = 0.047].
Latency (C)
The latency to leave the circle was non-significant [F (4, 45) = 0.86; p > 0.05]; circling behavior was absent (not shown).
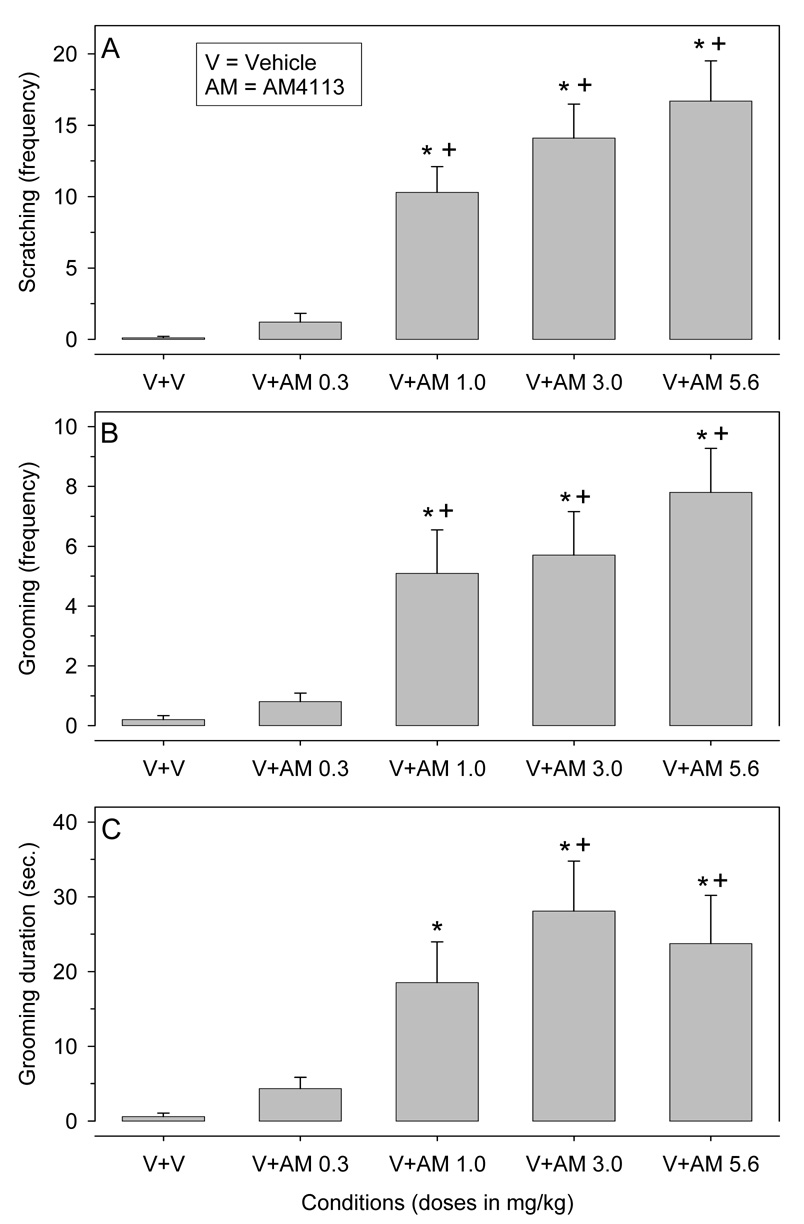
Figure 4 shows the effects of AM4113 (0, 0.3, 1, 3, and 5.6 mg/kg) alone on scratching (A), grooming frequency (B), and grooming duration (C).
Figure 4.
The effects of AM4113 (0, 0.3, 1, 3, and 5.6 mg/kg) on scratching (A), grooming frequency (B), and grooming duration (C) in different groups of Sprague-Dawley rats (n = 10). AM4113 (AM) and vehicle (V) injections were given i.p. 30 minutes prior to session onset; controls received two vehicle (2 ml/kg each) injections (V+V). The data points represent the means (±SEM) during a 5-minutes observation period in an open-field arena (n = 10).
Scratching frequency (A): *) different from controls (V+V); +) V+AM0.3 different from V+AM1, V+AM3 and V+AM5.6.
Grooming frequency (B): *) different from controls (V+V); +) V+AM0.3 different from V+AM1, V+AM3 and V+AM5.6.
Grooming duration (C): *) different from controls (V+V); +) V+AM0.3 different from V+AM3 and V+AM5.6.
Scratching (A)
AM4113 doses of 1, 3 and 5.6 mg/kg significantly increased the frequency of scratching compared to both the controls (V+V) and the low dose AM4113 condition (V+AM0.3) [F (4, 45) = 29.63; p < 0.001].
Grooming frequency (B)
AM4113 doses of 1, 3 and 5.6 mg/kg significantly increased the frequency of grooming compared to both the controls (V+V) and the low dose AM1346 condition (V+AM0.3) [F (4, 45) = 13.65; p < 0.001].
Grooming duration (C)
Doses of 1, 3 and 5.6 mg/kg of AM4113 resulted in significantly increased grooming duration compared to the controls (V+V). Significance was also evident when comparing the low dose condition (V+AM0.3) with the AM4113 doses of 3 (V+AM3) and 5.6 (V+AM5.6) mg/kg; [F (4, 45) = 8.82; p < 0.001].
Defecation, Urination and Vocalization (before and after) were all non-significant when examining AM4113 alone (p > 0.05; not shown).
4. Discussion
The current open-field studies were undertaken to determine if the putative CB1R neutral antagonist AM4113 would produce a different behavioral/open-field profile relative to rimonabant with particular emphasis on its effects regarding scratching and grooming. The latter behaviors showed dose-dependent increases after rimonabant administration in rats in our previous open-field studies (see also Navarro et al., 1997; Rubino et al., 1998; Tallett et al., 2007a; Vickers et al., 2003). To examine in vivo antagonism by AM4113 of drug agonist induced CB1R activation, the commonly employed CB1R ligand WIN was used.
Previously published results from our laboratory have demonstrated that rimonabant and AM251 are 143 and 430 times more selective for the CB1 than the CB2 receptors, respectively (Lan et al., 1999; McLaughlin et al., 2006). Other studies showed that the two compounds inhibit GTPγS (Landsman et al., 1997; McLaughlin et al., 2006) and increase cAMP accumulation (Mato et al., 2002; McLaughlin et al., 2006) and therefore can be considered CB1R inverse agonists. AM4113 is also CB1R selective with 100 fold selectivity over the CB2 receptor (Chambers et al., 2007; Sink et al., 2007), however it displays neutral antagonism in the cAMP assay as there was no change in the forskolin-induced cAMP levels using concentrations ranging from 630 nM to 10 µM of the compound as determined separately in two different assays (Chambers et al., 2007; Sink et al., 2007).
As expected, WIN (3 mg/kg) depressed ambulation and rearing (see e.g., Järbe et al., 2006). The WIN induced suppression of ambulation was antagonized by AM4113. The WIN induced suppression of rearing was clearly attenuated by AM4113 as evidenced by a significant increase in the frequency of rearing compared to that observed after administration of WIN alone. However, complete restoration of rearing to control levels was not achieved as evidenced by significant differences between controls and drug treated animals for all three antagonist doses given together with WIN. This has generally been the case also in our previous open-field studies involving higher doses of CB1R agonists (THC, methanandamide, AM411; WIN; and AM1346) and rimonabant (Järbe et al., 2002; 2003; 2004; 2006; 2007). The notion that complete restoration of behaviors suppressed by high doses of CB1R agonists may be difficult to achieve has been noted also by other investigators (McMahon et al., 2005 and references cited therein; see also Darmani, 2001), raising the possibility that not all behavioral effects following cannabinoid agonist administration are mediated through activation of CB1R only.
Circling occurred after administration of WIN (see Figure 2-C). Circling is a motor disturbance occurring after administration of higher doses of various CB1R agonists including THCs (Δ8-THC and Δ9-THC), cannabinol, AM411, HU210, AM1346 (Sjödén et al., 1973; Järbe and Hiltunen, 1987; Järbe et al., 2002; 2004; 2007; Ferrari et al., 1999) as well as WIN (Järbe et al., 2006; current study), but not methanandamide (Järbe et al., 1998; 2003). Circling as well as the increased latency to leave the middle start area of the open-field arena after WIN administration was antagonized by AM4113, which is consistent with previous findings using the above mentioned CB1R agonists and rimonabant. Additionally, no consistent patterns in the direction (i.e., rotating left or right) of the WIN induced circling behavior were evident which is in accord with previous data (Järbe et al., 2006; see also Järbe et al., 2002).
Scratching and grooming levels were low in the animals treated with WIN alone, an outcome characteristic of CB1R agonist administration in rats. Addition of AM4113 increased the levels of scratching and grooming in a dose-dependent fashion. Such an outcome is similar to what we have observed in previous open-field studies involving various CB1R agonists and rimonabant (Järbe et al., 2002; 2003; 2004; 2006; 2007). In addition to counteracting in vivo effects of WIN (current study), AM4113 also attenuated the effects of the CB1R agonist AM411 in a tetrad test (locomotion, analgesia, catalepsy and hypothermia) as described by Sink et al. (2007; see also Bergman et al., 2008) and AM4113 also blocked CP55,940 induced hypothermia, a measure of CNS activity (Chambers et al., 2007). Additionally, preliminary data by Bergman et al. (2008) suggested that effects induced by the potent CB1R agonist AM4054 were also blocked by AM4113, using hypothermia in rats and monkeys as the end-point. Thus, there is generality of the in vivo antagonism by AM4113 across structurally diverse CB1R agonists employing different behavioral end-points.
Administration of AM4113 alone (summarized in Figure 3 and Figure 4) tended to reduce ambulation (and possibly also rearing), but not the latency of the rats to leave the middle circle with all four feet (see also Sink et al., 2007). This behavioral pattern is in accord with our previous open-field studies involving rimonabant when examined alone; also, like rimonabant, AM4113 did not induce circling (Järbe et al., 2002; 2006). The increased incidence of scratching and grooming (both the frequency and duration) is also similar to our previously described data for rimonabant when given alone to rats (Järbe et al., 2002; 2006; see also Navarro et al., 1997; Pavón et al., 2006; Rubino et al., 1998; 2000; Tallett et al., 2007a; Vickers et al., 2003). Indeed, itching (pruritus) was cited as a reason for patients to discontinue taking the CB1R inverse agonist taranabant (Kirkham 2008), developed by Merck Pharmaceuticals as an anti-obesity agent (MK-0364). It remains to be determined if this would be a clinically significant issue should neutral CB1R antagonists be evaluated in humans.
Although the current studies did not detect a qualitative difference in vivo between the putative neutral antagonist AM4113 and the prototypical CB1R antagonist/inverse agonist rimonabant examining a variety of open-field generated behaviors, Chambers et al. (2007) reported that morphine-6-glucoronide-induced emesis in the ferret was significantly augmented by the rimonabant derivative AM251 but not by AM4113. Similarly, in the same dose range that suppressed feeding (Sink et al., 2007), AM4113 did not induce conditioned gaping (Salamone et al., 2007; Sink et al., 2007). Conditioned gaping is considered indicative of nausea in non-vomiting animals such as rats (Parker and Limebeer, 2006). In congruence with above data for ferrets (Chambers et al., 2007), AM251 produced conditioned gaping in rats (McLaughlin et al. 2005). Along the same vein, rimonabant augmented conditioned rejection reactions induced by lithium chloride in rats (Parker et al., 2003; see also De Vry et al., 2004), was emetic in the least shrew (Darmani, 2001), and associated with nausea in humans (Despres et al., 2005). Hence neutral antagonists, of which AM4113 may be an example, might afford a better treatment strategy for combating obesity compared to earlier developed CB1R antagonists/inverse agonists (Vemuri et al., 2008). However, our data also clearly show that AM4113 is not devoid of intrinsic activity (see also Salamone et al., 2007; Sink et al., 2007; 2008; Bergman et al., 2008). Like rimonabant, AM4113 produced dose-dependent increases in scratching and grooming. It is tempting to speculate that this intrinsic activity of the CB1R antagonists may be due to a change in endocannabinoid tone resulting from the CB1R blockade although other possibilities (e.g., antagonism of endogenously released adenosine at A1 receptors) also exist (Pertwee 2005). The effect of CB1R antagonists on motor activity may be due to mechanisms unrelated to those sub serving increased scratching and grooming, or alternatively, simply is a consequence of the increased rates of scratching and grooming. Indeed, response competition was proposed to be a confounding factor when evaluating acute feeding studies in rats (Tallett et al., 2007a, b). However, this probably cannot explain the reduced body weight sustained by continued administration of CB1R antagonists as tolerance develops to scratching and grooming after repeated rimonabant administration (Vickers et al., 2003).
In conclusion, the current results and above cited data suggest that the novel ligand AM4113 exert effects of its own that are qualitatively similar to those induced by rimonabant and other CB1R inverse agonists. Yet, unlike rimonabant and AM251, AM4113 did not induce signs indicative of nausea at doses effective in reducing food-intake in rodents, suggesting a potential benefit for CB1R neutral antagonists in the clinical management of obesity. Additional research is required to determine if the two classes of ligands (CB1R neutral and inverse agonists) diverge with regard to other end-points as well. Of special interest would be “mood alterations” (particularly anxiety and depression) associated with rimonabant administration resulting in reduced patient compliance (Christensen et al., 2007).
Acknowledgments
We thank Dr. Christopher S. Stearn for his comments on an earlier version of this manuscript. Supported by NIDA grants DA 09064, DA 00253 (TUCJ), and NIDA grants DA 03801, DA 07215, DA 09158 and DA 00493 (AM).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Bass CE, Griffin G, Grier M, Mahadevan A, Razdan RK, Martin BR. SR-141716A-induced stimulation of locomotor activity. A structure-activity relationship study. Pharmacol Biochem Behav. 2002;74:31–40. doi: 10.1016/s0091-3057(02)00945-0. [DOI] [PubMed] [Google Scholar]
- Bergman J, Delatte MS, Paronis CA, Vemuri K, Thakur GA, Makriyannis A. Some effects of CB1 antagonists with inverse agonist and neutral biochemical properties. Physiol Behav. 2008;93:666–670. doi: 10.1016/j.physbeh.2007.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canals M, Milligan G. Constitutive activity of the cannabinoid CB1 receptor regulates the function of co-expressed Mu opioid receptors. J Biol Chem. 2008;283:11424–11434. doi: 10.1074/jbc.M710300200. [DOI] [PubMed] [Google Scholar]
- Chambers AP, Vemuri VK, Peng Y, Wood JT, Olszewska T, Pittman QJ, Makriyannis A, Sharkey KA. A neutral CB1 receptor antagonist reduces weight gain in rat. Am J Physiol Regul Integr Comp Physiol. 2007;293:R2185–R2193. doi: 10.1152/ajpregu.00663.2007. [DOI] [PubMed] [Google Scholar]
- Christensen R, Kristensen PK, Bartels EM, Bliddal H, Astrup A. Efficacy and safety of the weight-loss drug rimonabant: a meta-analysis of randomised trials. Lancet. 2007;370:1706–1713. doi: 10.1016/S0140-6736(07)61721-8. [DOI] [PubMed] [Google Scholar]
- Compton DR, Aceto MD, Lowe J, Martin BR. In vivo characterization of a specific cannabinoid receptor antagonist (SR141716A): inhibition of delta-9-tetrahydrocannabinol-induced responses and apparent agonist activity. J Pharmacol Exp Ther. 1996;277:586–594. [PubMed] [Google Scholar]
- Cosenza M, Gifford AN, Gatley SJ, Pyatt B, Liu Q, Makriyannis A, Volkow ND. Locomotor activity and occupancy of brain cannabinoid CB1 receptors by the antagonist/inverse agonist AM281. Synapse. 2000;38:477–482. doi: 10.1002/1098-2396(20001215)38:4<477::AID-SYN13>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Darmani NA. The cannabinoid CB1 receptor antagonist SR 141716A reverses the antiemetic and motor depressant actions of WIN 55, 212-2. Eur J Pharmacol. 2001;430:49–58. doi: 10.1016/s0014-2999(01)01355-3. [DOI] [PubMed] [Google Scholar]
- Darmani NA, Pandya DK. Involvement of other neurotransmitters in behaviors induced by the cannabinoid CB1 receptor antagonist SR 141716A in naïve mice. J Neural Transm. 2000;107:931–945. doi: 10.1007/s007020070043. [DOI] [PubMed] [Google Scholar]
- Despres JP, Golay A, Sjostrom L. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med. 2005;353:2121–2134. doi: 10.1056/NEJMoa044537. [DOI] [PubMed] [Google Scholar]
- De Vry J, Schreiber R, Eckel G, Jentzsch KR. Behavioral mechanisms underlying inhibition of food-maintained responding by the cannabinoid receptor antagonist/inverse agonist SR 141716A. Eur J Pharmacol. 2004;483:55–63. doi: 10.1016/j.ejphar.2003.10.012. [DOI] [PubMed] [Google Scholar]
- Eilam D, Talangbayan H, Canaran G, Szechtman H. Dopaminergic control of locomotion, mouthing, snout contact, and grooming: opposing roles of D1 and D2 receptors. Psychopharmacology (Berl) 1992;106:447–454. doi: 10.1007/BF02244813. [DOI] [PubMed] [Google Scholar]
- Felder CC, Joyce KE, Briley EM, Glass M, Mackie KP, Fahey KJ, Cullinan GJ, Hunden DC, Johnson DW, Chaney MO, Koppel GA, Brownstein M. LY320135, a novel cannabinoid CB1 receptor antagonist, unmasks coupling of the CB1 receptor to stimulation of cAMP accumulation. J Pharmacol Exp Ther. 1998;284:291–297. [PubMed] [Google Scholar]
- Ferrari F, Ottani A, Giuliani D. Cannabinoid activity in rats and pigeons of HU 210, a potent anti-emetic drug. Pharmacol Biochem Behav. 1999;62:75–80. doi: 10.1016/s0091-3057(98)00114-2. [DOI] [PubMed] [Google Scholar]
- Fride E, Braun H, Matan H, Steinberg S, Reggio PH, Seltzman HH. Inhibition of milk ingestion and growth after administration of a neutral cannabinoid CB1 receptor antagonist on the first postnatal day in the mouse. Pediatr Res. 2007;62:533–536. doi: 10.1203/PDR.0b013e3181559d42. [DOI] [PubMed] [Google Scholar]
- Gardner A, Mallet PE. Suppression of feeding, drinking, and locomotion by a putative cannabinoid receptor 'silent’ antagonist. Eur J Pharmacol. 2006;530:103–106. doi: 10.1016/j.ejphar.2005.11.032. [DOI] [PubMed] [Google Scholar]
- Govaerts SJ, Muccioli GG, Hermans E, Lambert DM. Characterization of the pharmacology of imidazolidinedione derivatives at cannabinoid CB1 and CB2 receptors. Eur J Pharmacol. 2004;495:43–53. doi: 10.1016/j.ejphar.2004.05.023. [DOI] [PubMed] [Google Scholar]
- Greasley PJ, Clapham JC. Inverse agonism or neutral antagonism at G-protein coupled receptors: a medicinal chemistry challenge worth pursuing? Eur J Pharmacol. 2006;553:1–9. doi: 10.1016/j.ejphar.2006.09.032. [DOI] [PubMed] [Google Scholar]
- Howlett AC, Breivogel CS, Childers SR, Deadwyler SA, Hampson RE, Porrino LJ. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology. 2004;47:345–358. doi: 10.1016/j.neuropharm.2004.07.030. [DOI] [PubMed] [Google Scholar]
- Hurst D, Umejiego U, Lynch D, Seltzman H, Hyatt S, Roche M, McAllister S, Fleischer D, Kapur A, Abood M, Shi S, Jones J, Lewis D, Reggio P. Biarylpyrazole inverse agonists at the cannabinoid CB1 receptor: importance of the C-3 carboxamide oxygen/lysine3.28(192) interaction. J Med Chem. 2006;49:5969–5987. doi: 10.1021/jm060446b. [DOI] [PubMed] [Google Scholar]
- Jagerovic N, Hernandez-Folgado L, Alkorta I, Goya P, Navarro M, Serrano A, Rodriguez de Fonseca F, Dannert MT, Alsasua A, Suardiaz M, Pascual D, Martin MI. Discovery of 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-3-hexyl-1h-1,2,4-triazole, a novel in vivo cannabinoid antagonist containing a 1,2,4-triazole motif. J Med Chem. 2004;47:2939–2942. doi: 10.1021/jm031099y. [DOI] [PubMed] [Google Scholar]
- Janoyan JJ, Crim JL, Darmani NA. Reversal of SR 141716A-induced head-twitch and ear-scratch responses in mice by Δ9-THC and other cannabinoids. Pharmacol Biochem Behav. 2002;71:155–162. doi: 10.1016/s0091-3057(01)00647-5. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, Andrzejewski ME, DiPatrizio NV. Interactions between the CB1 receptor agonist Δ9-THC and the CB1 receptor antagonist SR-141716 in rats: open-field revisited. Pharmacol Biochem Behav. 2002;73:911–919. doi: 10.1016/s0091-3057(02)00938-3. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, DiPatrizio NV, Li C, Makriyannis A. The cannabinoid receptor antagonist SR-141716 does not readily antagonize open-field effects induced by the cannabinoid receptor agonist (R)-methanandamide in rats. Pharmacol Biochem Behav. 2003;75:809–821. doi: 10.1016/s0091-3057(03)00168-0. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, DiPatrizio NV, Li C, Makriyannis A. Effects of AM1346, a high-affinity CB1 receptor selective anandamide analog, on open-field behavior in rats. Behav Pharmacol. 2007;18:673–680. doi: 10.1097/FBP.0b013e3282f00bbf. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, DiPatrizio NV, Lu D, Makriyannis A. (-)-Adamantyl-Δ8-tetrahydrocannabinol (AM-411), a selective cannabinoid CB1 receptor agonist: effects on open-field behaviors and antagonism by SR-141716 in rats. Behav Pharmacol. 2004;15:517–521. doi: 10.1097/00008877-200411000-00008. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, Hiltunen AJ. Cannabimimetic activity of cannabinol in rats and pigeons. Neuropharmacology. 1987;26:219–228. doi: 10.1016/0028-3908(87)90212-7. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, Ross T, DiPatrizio NV, Pandarinathan L, Makriyannis A. Effects of the CB1R agonist WIN and the CB1R antagonists SR-141716 and AM-1387: an open-field examination in rats. Pharmacol Biochem Behav. 2006;85:243–252. doi: 10.1016/j.pbb.2006.08.006. [DOI] [PubMed] [Google Scholar]
- Järbe TUC, Sheppard R, Lamb RJ, Makriyannis A, Lin S, Goutopoulos A. Effects of delta-9-THC and (R)-methanandamide on open-field behaviors in rats. Behav Pharmacol. 1998;9:169–174. [PubMed] [Google Scholar]
- Kirk RE. Experimental design: procedures for the behavioral sciences. Belmont (CA): Brooks/Cole; 1968. [Google Scholar]
- Kirkham TC. Taranabant cuts the fat: new hope for cannabinoid-based obesity therapies? Cell Metab. 2008;7:1–2. doi: 10.1016/j.cmet.2007.12.006. [DOI] [PubMed] [Google Scholar]
- Lan R, Liu Q, Fan P, Lin S, Fernando SR, McCallion D, Pertwee R, Makriyannis A. Structure-activity relationships of pyrazole derivatives as cannabinoid receptor antagonists. J Med Chem. 1999;42:769–776. doi: 10.1021/jm980363y. [DOI] [PubMed] [Google Scholar]
- Landsman RS, Burkey TH, Consroe P, Roeske WR, Yamamura HI. SR141716A is an inverse agonist at the human cannabinoid CB1 receptor. Eur J Pharmacol. 1997;334:R1–R2. doi: 10.1016/s0014-2999(97)01160-6. [DOI] [PubMed] [Google Scholar]
- Martin BR, Stevenson LA, Pertwee RG, Breivogel CS, Wi1liams W, Mahadevan A, Razdan RK. Agonists and silent antagonists in a series of cannabinoids sulfonamides. The International Cannabinoid Research Society; 2002. [Google Scholar]
- Mato S, Pazos A, Valdizan EM. Cannabinoid receptor antagonism and inverse agonism in response to SR141716A on cAMP production in human and rat brain. Eur J Pharmacol. 2002;443:43–46. doi: 10.1016/s0014-2999(02)01575-3. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Winston KM, Limebeer CL, Parker LA, Makriyannis A, Salamone JD. The cannabinoid CB1 antagonist AM 251 produces food avoidance and behaviors associated with nausea but does not impair feeding efficiency in rats. Psychopharmacology (Berl) 2005;180:286–293. doi: 10.1007/s00213-005-2171-0. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Qian L, Wood JT, Wisniecki A, Winston KM, Swezey LA, Ishiwari K, Betz AJ, Pandarinathan L, Xu W, Makriyannis A, Salamone JD. Suppression of food intake and food-reinforced behavior produced by the novel CB1 receptor antagonist/inverse agonist AM 1387. Pharmacol Biochem Behav. 2006;83:396–402. doi: 10.1016/j.pbb.2006.02.022. [DOI] [PubMed] [Google Scholar]
- McMahon LR, Amin MR, France CP. SR 141716A differentially attenuates the behavioral effects of Δ9-THC in rhesus monkeys. Behav Pharmacol. 2005;16:363–372. doi: 10.1097/00008877-200509000-00008. [DOI] [PubMed] [Google Scholar]
- National Institutes of Health. Washington, D.C.: National Academy Press; Principles of Animal Laboratory Care. 1996
- Navarro M, Hernandez E, Munoz RM, del Arco I, Villanua MA, Carrera MR, Rodriguez de Fonseca F. Acute administration of the CB1 cannabinoid receptor antagonist SR 141716A induces anxiety-like responses in the rat. Neuroreport. 1997;8:491–496. doi: 10.1097/00001756-199701200-00023. [DOI] [PubMed] [Google Scholar]
- Parker LA, Limebeer CL. Conditioned gaping in rats: a selective measure of nausea. Auton Neurosci. 2006;129:36–41. doi: 10.1016/j.autneu.2006.07.022. [DOI] [PubMed] [Google Scholar]
- Parker LA, Mechoulam R, Schlievert C, Abbott L, Fudge ML, Burton P. Effects of cannabinoids on lithium-induced conditioned rejection reactions in a rat model of nausea. Psychopharmacology (Berl) 2003;166:156–162. doi: 10.1007/s00213-002-1329-2. [DOI] [PubMed] [Google Scholar]
- Pavón FJ, Bilbao A, Hernández-Folgado L, Cippitelli A, Jagerovic N, Abellan G, Rodriguez-Franco MA, Serrano A, Macías M, Gomez R, Navarro M, Goya P, de Fonseca FR. Antiobesity effects of the novel in vivo neutral cannabinoid receptor antagonist 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-3-hexyl-1H-1,2,4-triazole - LH 21. Neuropharmacology. 2006;51:358–366. doi: 10.1016/j.neuropharm.2006.03.029. [DOI] [PubMed] [Google Scholar]
- Pavón FJ, Serrano A, Pérez-Valero V, Jagerovic N, Hernández-Folgado L, Bermúdez-Silva FJ, Macías M, Goya P, de Fonseca FR. Central versus peripheral antagonism of cannabinoid CB1 receptor in obesity: effects of LH-21, a peripherally acting neutral cannabinoid receptor antagonist, in Zucker rats. J Neuroendocrinol. 2008 Suppl 1:116–123. doi: 10.1111/j.1365-2826.2008.01693.x. [DOI] [PubMed] [Google Scholar]
- Pertwee RG. Inverse agonism and neutral antagonism at cannabinoid CB1 receptors. Life Sci. 2005;76:1307–1324. doi: 10.1016/j.lfs.2004.10.025. [DOI] [PubMed] [Google Scholar]
- Rinaldi-Carmona M, Barth F, Héaulme M, Shire D, Calandra B, Congy C, Martinez S, Maruani J, Néliat G, Caput D, Ferrara P, Soubrié P, Breliére JC, LeFur G. SR 141716A, a potent and selective antagonist of the brain cannabinoid receptor. FEBS Lett. 1994;350:240–244. doi: 10.1016/0014-5793(94)00773-x. [DOI] [PubMed] [Google Scholar]
- Rinaldi-Carmona M, Barth F, Congy C, Martinez S, Oustric D, Perio A, Poncelet M, Maruani J, Arnone M, Finance O, Soubrie P, LeFur G. SR147778 [5-(4-bromophenyl)-1-(2,4-dichlorophenyl)-4-ethyl-N-(1-piperidinyl)-1H-pyrazole-3-carboxamide], a new potent and selective antagonist of the CB1 cannabinoid receptor: biochemical and pharmacological characterization. J Pharmacol Exp Ther. 2004;310:905–914. doi: 10.1124/jpet.104.067884. [DOI] [PubMed] [Google Scholar]
- Rodriguez de Fonseca F, Del Arco I, Martin-Calderon JL, Gorriti MA, Navarro M. Role of the endogenous cannabinoid system in the regulation of motor activity. Neurobiol Dis. 1998;5(6 Pt B):483–501. doi: 10.1006/nbdi.1998.0217. [DOI] [PubMed] [Google Scholar]
- Rubino T, Patrini G, Massi P, Fuzio D, Vigano D, Giagnoni G, Parolaro D. Cannabinoid-precipitated withdrawal: a time-course study of the behavioral aspect and its correlation with cannabinoid receptors and G protein expression. J Pharmacol Exp Ther. 1998;285:813–819. [PubMed] [Google Scholar]
- Rubino T, Vigano D, Zagato E, Sala M, Parolaro D. In vivo characterization of the specific cannabinoid receptor antagonist, SR141716A: behavioral and cellular responses after acute and chronic treatments. Synapse. 2000;35:8–14. doi: 10.1002/(SICI)1098-2396(200001)35:1<8::AID-SYN2>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Ruiu S, Pinna GA, Marchese G, Mussinu JM, Saba P, Tambaro S, Casti P, Vargiu R, Pani L. Synthesis and characterization of NESS 0327: a novel putative antagonist of the CB1 cannabinoid receptor. J Pharmacol Exp Ther. 2003;306:363–370. doi: 10.1124/jpet.103.049924. [DOI] [PubMed] [Google Scholar]
- Salamone JD, McLaughlin PJ, Sink K, Makriyannis A, Parker LA. Cannabinoid CB1 receptor inverse agonists and neutral antagonists: effects on food intake, food-reinforced behavior and food aversions. Physiol Behav. 2007;91:383–388. doi: 10.1016/j.physbeh.2007.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sink KS, McLaughlin PJ, Wood JA, Brown C, Fan P, Vemuri VK, Pang Y, Olzewska T, Thakur GA, Makriyannis A, Parker LA, Salamone JD. The novel cannabinoid CB(1) receptor neutral antagonist AM4113 suppresses food intake and food-reinforced behavior but does not induce signs of nausea in rats. Neuropsychopharmacology. 2008;33:946–955. doi: 10.1038/sj.npp.1301476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sink KS, Vemuri VK, Olszewska T, Makriyannis A, Salamone JD. Cannabinoid CB1 antagonists and dopamine antagonists produce different effects on a task involving response allocation and effort-related choice in food-seeking behavior. Psychopharmacology (Berl) 2008;196:565–574. doi: 10.1007/s00213-007-0988-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sjödén P-O, Järbe TUC, Henriksson BG. Effects of long-term administration and withdrawal of tetrahydrocannabinols (delta-8-THC and delta-9-THC) on open-field behavior in rats. Pharmacol Biochem Behav. 1973;1:243–249. doi: 10.1016/0091-3057(73)90111-1. [DOI] [PubMed] [Google Scholar]
- Tallett AJ, Blundell JE, Rodgers RJ. Grooming, scratching and feeding: role of response competition in acute anorectic response to rimonabant in male rats. Psychopharmacology (Berl) 2007a;195:27–39. doi: 10.1007/s00213-007-0880-2. [DOI] [PubMed] [Google Scholar]
- Tallett AJ, Blundell JE, Rodgers JR. Acute anorectic response to cannabinoid CB1 receptor antagonist/inverse agonist AM 251 in rats: indirect behavioural mediation. Behav Pharmacol. 2007b;18:591–600. doi: 10.1097/FBP.0b013e3282eff0a9. [DOI] [PubMed] [Google Scholar]
- Thakur GA, Nikas SP, Li C, Makriyannis A. Structural requirements for cannabinoid receptor probes. In: Pertwee R, editor. Handbook of experimental pharmacology. Volume 168. New York, NY: Springer; 2005. pp. 209–246. [DOI] [PubMed] [Google Scholar]
- Vemuri VK, Janero DR, Makriyannis A. Pharmacotherapeutic targeting of the endocannabinoid signaling system: drugs for obesity and the metabolic syndrome. Physiol Behav. 2008;93:671–686. doi: 10.1016/j.physbeh.2007.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickers SP, Webster LJ, Wyatt A, Dourish CT, Kennett GA. Preferential effects of the cannabinoid CB1 receptor antagonist, SR 141716, on food intake and body weight gain of obese (fa/fa) compared to lean Zucker rats. Psychopharmacology (Berl) 2003;167:103–111. doi: 10.1007/s00213-002-1384-8. [DOI] [PubMed] [Google Scholar]