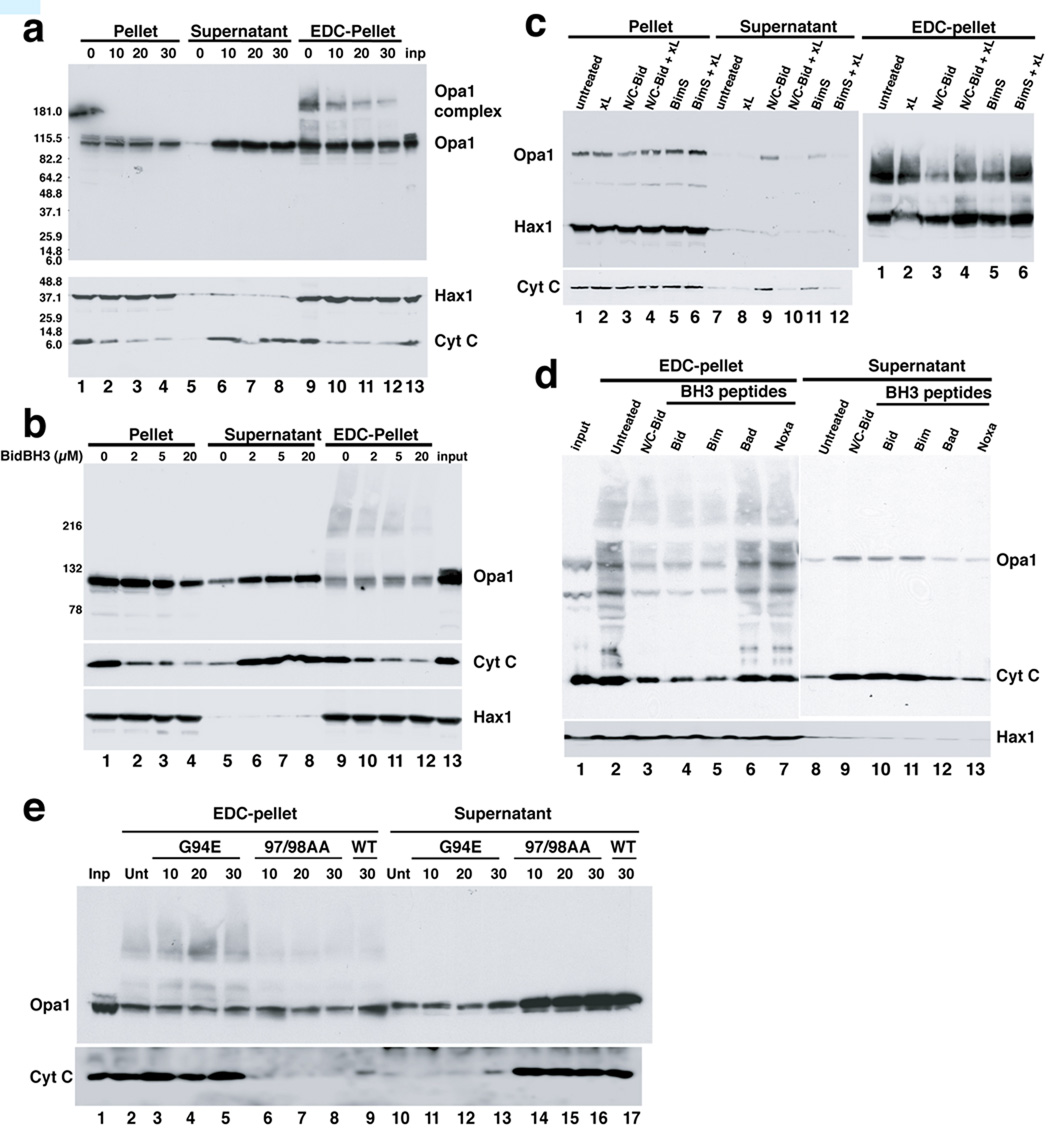
Fig. 1. Disassembly of OPA1 complexes by BH3-only proteins and their corresponding peptides.
a. N/C-Bid induced cytochrome c release and OPA1 complex disassembly, with similar kinetics. Isolated mitochondria were suspended in trehalose buffer at 5 mg/ml and incubated with 10 nM N/C-Bid for 0,10, 20 and 30 min. at 37° C. Samples were centrifuged, and pellets were either suspended right away in SDS sample buffer or treated for 45 minutes in 10 mM EDC (Pierce) in PBS at 37° C before the addition of SDS buffer. Proteins in the pellets and supernatants were separated by 4–12% polyacrylamide gradient gels and analyzed by immunoblotting using anti-OPA1, HAX1 and cytochrome c antibodies. b. Bid BH3 peptide produced similar effects. Mitochondrial samples were incubated either with 0–20 µM of BH3 peptide derived from Bid or left untreated for 30 min. c. BimS protein causes a similar disassembly of OPA1 complex, which is inhibited by Bcl-xL. Samples were treated with 10 nM N/C-Bid, 40 nM BimS, 200 nM Bcl-xLΔC, either alone or together for 30 minutes. d. OPA1 disassembly was induced by Bid and Bim BH3 peptides, but not other BH3 peptides. Samples were treated either with 10 nM Bid protein or 20 µM BH3 peptides derived from Bid, Bim, Bad or Noxa or left untreated for 30 min. e. BH3 domain of tBid is necessary for cytochrome c release and disassembly of OPA1 complex. Isolated mitochondria were treated with either 10 nM N/C Bid or its mutants G94E and M97AD98A for 10–30 min. at 37° C.