Figure 5.
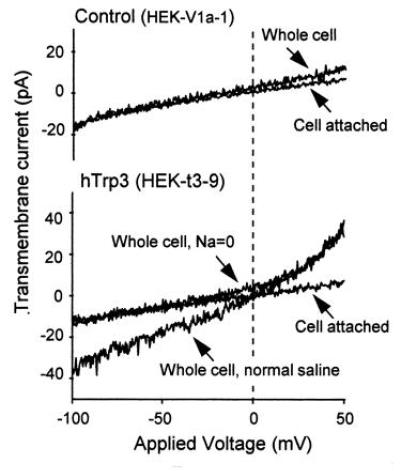
Development of a nonselective cation current in HEK 293 cells expressing human Trp3 after loading with BAPTA. HEK cells were transfected with pcDNA3 carrying either the HA epitope-tagged Trp3 cDNA (hTrp3), or as control, the human type-2 vasopressin receptor cDNA (V2R) using the calcium phosphate/glycerol shock method. G418-resistant colonies were selected and tested for expression of the respective inserts—the HA epitope by immunocytochemistry and the V2R by its ability to stimulate adenylyl cyclase. Clonal cell lines were then isolated by limiting dilution, retested for expression of the proteins, and tested for ionic transmembrane currents using the whole-cell patch clamp technique. The pipette contained 150 mM Cs-aspartate, 4 mM MgCl2, 12 mM Na-BAPTA, and 10 mM Hepes. The extracellular medium contained normal saline: 155 mM NaCl, 2 mM KCl, 2 mM MgCl2, 10 mM glucose, and 10 mM Hepes. Na-free saline contained: 110 mM N-methylglucammonium (NMG)-Mes, 5 mM H-EDTA, 4.3 mM CaCl2, and 10 mM Hepes (free Ca2+, 20 μM). The cells were subjected to a depolarizing ramp from −150 mV to +150 mV. The upper portion shows the records of a control V2R-positive cell; the lower portion shows transmembrane currents of a cell expressing hTrp3. Note presence in the hTrp3-expressing cells of a nonselective conductance that is absent in control HEK cells as well as in the cell attached patch before establishing the whole cell configuration. Exchanging NMG+ for Na+ abolished the inward aspect of the transmembrane current but did not interfere with the outward current carried by Cs+ (data not shown). Other experiments showed that the channel is permeable to Ca2+ as well as Na+.
