Abstract
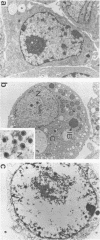
We studied the susceptibility of mouse testicular interstitial Leydig cells to cytomegalovirus both in vivo and in vitro. The in vivo studies included intratesticular and intraperitoneal infection of 6-week-old mice with murine cytomegalovirus (MCMV); the in vitro studies involved an MCMV-Leydig cell interaction using a Leydig tumor cell line (I-10). MCMV-specific antigens were detected in interstitial Leydig cells in sections of MCMV-inoculated testes by an indirect immunofluorescence test. MCMV DNA was also localized in the same testes cells derived from mice, which received intratesticular and intraperitoneal MCMV inoculations, respectively, by in situ DNA-RNA hybridization. Cytopathic effects were seen in MCMV-infected I-10 cell cultures 2 or more days after exposure to MCMV. The infected cells showed intranuclear inclusions characteristic of cytomegalovirus when stained with May-Grunwald-Giemsa stain. The indirect immunofluorescence test was also positive with MCMV-infected I-10 cells. MCMV DNA was detected in these cells by in situ DNA-RNA cytohybridization, and the presence of viral particles in MCMV-infected I-10 cells was confirmed by electron microscopy. Thus, we conclude that the interstitial Leydig cell is susceptible to MCMV infection both in vivo and in vitro.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baskar J. F., Huang E. S. Effect of murine cytomegalovirus on implantation stage mouse embryos. Proc Soc Exp Biol Med. 1981 Sep;167(4):581–587. doi: 10.3181/00379727-167-41219. [DOI] [PubMed] [Google Scholar]
- Biggers J. D., Baskar J. F., Torchiana D. F. Reduction of fertility of mice by the intrauterine injection of prostaglandin antagonists. J Reprod Fertil. 1981 Nov;63(2):365–372. doi: 10.1530/jrf.0.0630365. [DOI] [PubMed] [Google Scholar]
- Brautigam A. R., Oldstone M. B. Replication of murine cytomegalovirus in reproductive tissues. Am J Pathol. 1980 Jan;98(1):213–224. [PMC free article] [PubMed] [Google Scholar]
- Davis G. L. Cytomegalovirus in the inner ear. Case report and electron microscopic study. Ann Otol Rhinol Laryngol. 1969 Dec;78(6):1179–1188. doi: 10.1177/000348946907800604. [DOI] [PubMed] [Google Scholar]
- Desjardins C. Endocrine signaling and male reproduction. Biol Reprod. 1981 Feb;24(1):1–21. doi: 10.1095/biolreprod24.1.1. [DOI] [PubMed] [Google Scholar]
- Diosi P., Babusceac L., Nevinglovschi O., Kun-Stoicu G. Cytomegalovirus infection associated with pregnancy. Lancet. 1967 Nov 18;2(7525):1063–1066. doi: 10.1016/s0140-6736(67)90338-8. [DOI] [PubMed] [Google Scholar]
- Dutko F. J., Oldstone M. B. Murine cytomegalovirus infects spermatogenic cells. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2988–2991. doi: 10.1073/pnas.76.6.2988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haseltine F. P., Ohno S. Mechanisms of gonadal differentiation. Science. 1981 Mar 20;211(4488):1272–1278. doi: 10.1126/science.7010601. [DOI] [PubMed] [Google Scholar]
- Huang E. S., Chen S. T., Pagano J. S. Human cytomegalovirus. I. Purification and characterization of viral DNA. J Virol. 1973 Dec;12(6):1473–1481. doi: 10.1128/jvi.12.6.1473-1481.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilpatrick B. A., Huang E. S., Pagano J. S. Analysis of cytomegalovirus genomes with restriction endonucleases Hin D III and EcoR-1. J Virol. 1976 Jun;18(3):1095–1105. doi: 10.1128/jvi.18.3.1095-1105.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar M. L., Nankervis G. A. Cytomegalovirus infections. South Med J. 1979 Jul;72(7):854–861. doi: 10.1097/00007611-197907000-00027. [DOI] [PubMed] [Google Scholar]
- Lang D. J., Kummer J. F. Demonstration of cytomegalovirus in semen. N Engl J Med. 1972 Oct 12;287(15):756–758. doi: 10.1056/NEJM197210122871508. [DOI] [PubMed] [Google Scholar]
- Lang D. J., Kummer J. F., Hartley D. P. Cytomegalovirus in semen. Persistence and demonstration in extracellular fluids. N Engl J Med. 1974 Jul 18;291(3):121–123. doi: 10.1056/NEJM197407182910303. [DOI] [PubMed] [Google Scholar]
- Montplaisir S., Belloncik S., Leduc N. P., Onji P. A., Martineau B., Kurstak E. Electron microscopy in the rapid diagnosis of cytomegalovirus: ultrastructural observation and comparison of methods of diagnosis. J Infect Dis. 1972 May;125(5):533–538. doi: 10.1093/infdis/125.5.533. [DOI] [PubMed] [Google Scholar]
- Naftolin F. Understanding the bases of sex differences. Science. 1981 Mar 20;211(4488):1263–1264. doi: 10.1126/science.7209509. [DOI] [PubMed] [Google Scholar]
- Numazaki Y., Yano N., Morizuka T., Takai S., Ishida N. Primary infection with human cytomegalovirus: virus isolation from healthy infants and pregnant women. Am J Epidemiol. 1970 Apr;91(4):410–417. doi: 10.1093/oxfordjournals.aje.a121151. [DOI] [PubMed] [Google Scholar]
- Rubin R. T., Reinisch J. M., Haskett R. F. Postnatal gonadal steroid effects on human behavior. Science. 1981 Mar 20;211(4488):1318–1324. doi: 10.1126/science.7209511. [DOI] [PubMed] [Google Scholar]
- Schumacher M., Schäfer G., Holstein A. F., Hilz H. Rapid isolation of mouse Leydig cells by centrifugation in Percoll density gradients with complete retention of morphological and biochemical integrity. FEBS Lett. 1978 Jul 15;91(2):333–338. doi: 10.1016/0014-5793(78)81204-6. [DOI] [PubMed] [Google Scholar]
- Shin S. I. Studies on interstitial cells in tissue culture: steroid biosynthesis in monolayers of mouse testicular interstitial cells. Endocrinology. 1967 Sep;81(3):440–448. doi: 10.1210/endo-81-3-440. [DOI] [PubMed] [Google Scholar]
- WELLER T. H., HANSHAW J. B. Virologic and clinical observations on cytomegalic inclusion disease. N Engl J Med. 1962 Jun 14;266:1233–1244. doi: 10.1056/NEJM196206142662401. [DOI] [PubMed] [Google Scholar]
- Wilson G. A., Young F. E. Isolation of a sequence-specific endonuclease (BamI) from Bacillus amyloliquefaciens H. J Mol Biol. 1975 Sep 5;97(1):123–125. doi: 10.1016/s0022-2836(75)80028-3. [DOI] [PubMed] [Google Scholar]
- Zain B. S., Roberts R. J. A new specific endonuclease from Xanthomonas badrii. J Mol Biol. 1977 Sep 15;115(2):249–255. doi: 10.1016/0022-2836(77)90101-2. [DOI] [PubMed] [Google Scholar]