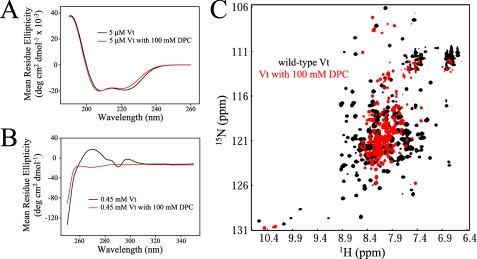
FIGURE 4.
CD and NMR analysis of Vt in the presence of DPC micelles. Comparison of far (A) and near (B) UV CD spectra of Vt in the presence and absence of 100 mm DPC. While far UV CD spectra show similar spectral profiles, significant alterations are observed in the near UV CD spectra. Results from these analyses indicate that DPC micelles cause a loss in Vt tertiary structure but maintain helical secondary structure. The data are further supported by NMR studies. 1H-15N HSQC NMR spectra were collected on 0.15 mm Vt at pH 5.5, both alone and in the presence of 100 mm DPC (C). The loss of spectral dispersion and the increase in resonances with chemical shifts close to the random coil values (∼8.3 ppm), suggest a loss in Vt tertiary structure upon association with DPC micelles. As vinculin does not appear to specifically bind PC, interactions with DPC micelles are likely to be nonspecific. In addition, similar structural perturbations were observed with all lipid micelles tested (i.e. LPPG and short-chain PIP2), suggesting that lipid micelles may act as a detergent, unfolding Vt.