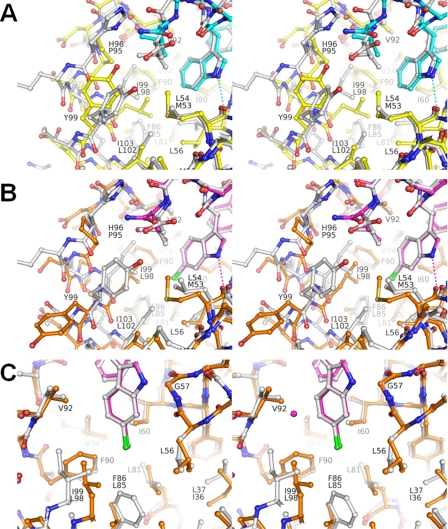
FIGURE 3.
HdmX and Hdm2 have important differences in the Leu and Trp pockets. A, superposition of the complexes HdmX-compound 1 (color coding is as in Figs. 1A and 2, i.e. HdmX with carbons are in yellow, compound 1 with carbons are in cyan) and Hdm2-optimized p53 peptide (PDB entry code 1T4F; Hdm2 and ligand with carbons in white) zoomed in on the Leu and Trp pockets. Amino acid residues that differ in identity between HdmX and Hdm2 have two labels (upper label from Hdm2), otherwise only one label (numbering for HdmX). The differences Pro95 versus His96, Met53 versus Leu54, and Leu98 versus Ile99 for HdmX versus Hdm2 modify the shape of the Leu pocket. In particular, the presence and position of CB-Pro95 for HdmX leads to a different position of the Leu side chain from the ligand. B, superposition of the complexes HdmX-compound 2 (color coding as in Fig. 2, i.e. HdmX with carbons in brown, compound 2 with carbons in magenta) and Hdm2-compound 2 (PDB entry code 2GV2; Hdm2 and ligand with carbons in white), zoomed in on the Leu and Trp pockets. The presence of a 6-chlorine substituent at the bottom of the Trp pocket leads to dramatic side chain movements of Leu98, Tyr99, and Pro95 for HdmX, where as Hdm2 shows practically no changes (cf. Figs. 2 and 3A). The Leu side chain of compound 2 can now adopt a very similar position for the HdmX and Hdm2 complexes (because the Leu pocket has been widened for HdmX), in contrast to the situation with a 6-H substituent in the Trp pocket (Fig. 3A). C, same superposition as in Fig. 3B but zoomed in on the Trp pocket. The differences Leu85 versus Phe86 and Leu98 versus Ile99 for HdmX versus Hdm2 modify the shape of the Trp pocket. In particular, the bottom of the Trp pocket is not yet completely filled by the 6-chlorine for HdmX. The important van der Waals interactions made by the 6-chlorine with Phe86 for Hdm2 are only partially substituted by hydrophobic interactions with Leu98 for HdmX. The diagram is programmed for stereo viewing.