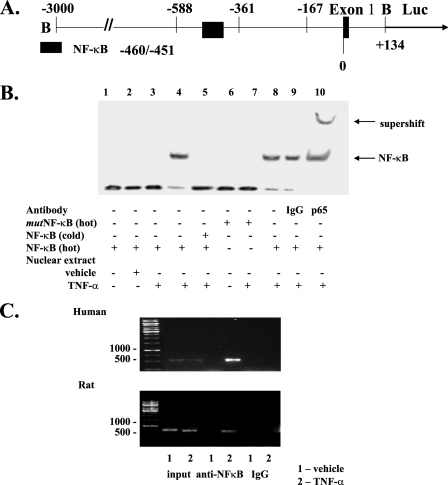
FIGURE 5.
NF-κB binds to its putative site in the S100A6 promoter in vitro and in vivo in response to TNF-α. A, schematic representation of the -3000/+134 S100A6 promoter fragment containing the first exon. The location of the restriction sites (B, BamHI) used to generate the fragment and location of the NF-κB site are shown. B, electromobility shift assay using nuclear extracts from myocyte cultures treated with TNF-α (5 ng/ml) or vehicle as indicated. The binding of NF-κB was confirmed by point mutations (mut) in NF-κB oligonucleotides, addition of 100-fold excess cold competitor oligonucleotides, and by supershift with an antibody to NF-κB p65 as indicated. C, myocyte cultures were co-transfected with the -588/+134 human S100A6 promoter-luciferase reporter construct as indicated and treated with TNF-α (5 ng/ml) or vehicle for 1 h then harvested and then subjected to chromatin immunoprecipitation analysis using NF-κB p65 antibody or a nonspecific anti-rabbit IgG. The isolated DNA fragments were amplified by PCR using both human (C, top) and rat (C, bottom) S100A6-specific primers. Input, nonprecipitated chromatin.