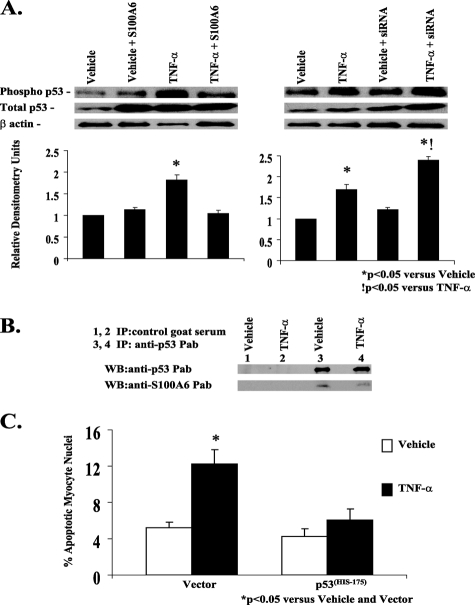
FIGURE 9.
S100A6 modulation of TNF-α-induced myocyte apoptosis requires p53. A, representative Western blot of phospho-p53, total p53, and β-actin in myocyte cultures transfected with either vector alone, human S100A6 expression plasmid, or an siRNA duplex against S100A6 and treated for 48 h with either vehicle or TNF-α (5 ng/ml). Respective relative densitometry units are displayed below blots. *, p < 0.05 versus vehicle and vector.!, p < 0.05 versus TNF-α and vector. n = 3. B, co-immunoprecipitation of p53 and S100A6 in TNF-α-treated myocyte cultures. Aliquots of lysates of myocyte cultures treated with either vehicle or TNF-α were incubated with control goat serum (lanes 1 and 2) or anti-p53 polyclonal antibody (lanes 3 and 4), followed by incubation with protein A-Sepharose. The immune complexes were dissociated and analyzed by Western blotting with anti-S100A6 or anti-p53 antibodies as indicated in each blot. C, myocyte cultures were co-transfected with a mutant p53His175 plasmid acting as a dominant negative and treated for 48 h with either TNF-α (5 ng/ml), 5% serum, or vehicle. The results are the mean ± S.E. of six different experiments. *, p < 0.05 versus vehicle/vector.