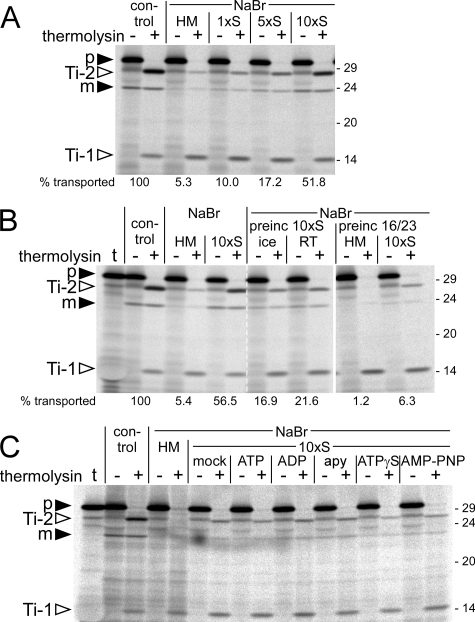
FIGURE 3.
Effect of stromal concentration on the transport properties of NaBr-extracted thylakoids. A, thylakoids were extracted with 1 m NaBr, resuspended in either HM buffer (HM) or stromal extract of different concentration (1xS, 5xS, 10xS) and subjected to thylakoid transport experiments analyzing the 16/23 precursor (p). The relative amounts of translocated protein (i.e. Ti-2 plus mature protein (m), taking into account the respective number of [35S]methionine residues) were quantified (in terms of the percentage of the control reaction) and are given below the corresponding lanes. B, thylakoids extracted with 1 m NaBr were preincubated for 10 min with 10-fold concentrated stromal extract (preinc 10xS) either on ice or at room temperature (RT) and washed once with HM buffer prior to the import reaction (middle panel). Alternatively, thylakoids extracted with 1 m NaBr were preincubated for 2 min on ice with the 16/23 precursor protein and subsequently washed once with HM buffer to remove excess precursor (preinc 16/23, right panel). Thylakoids were then resuspended in either HM buffer or 10-fold concentrated stroma (10xS) upon starting the import reaction. In the left panel, import experiments analogous to that shown in A were performed in parallel for comparison. Quantification of the signal intensities was carried out as in A. In lanes t, 1 μl of the respective in vitro translation assay was loaded. C, thylakoids extracted with 1 m NaBr were resuspended in either HM buffer or 10-fold concentrated stromal extract that was further supplemented with buffer (mock), 5 mm ATP, 5 mm ADP, 2 units of apyrase (apy), 5 mm ATPγS, or 5 mm AMP-PNP and subjected to thylakoid transport assays using the 16/23 precursor protein. For further details, see the legend for Fig. 1.