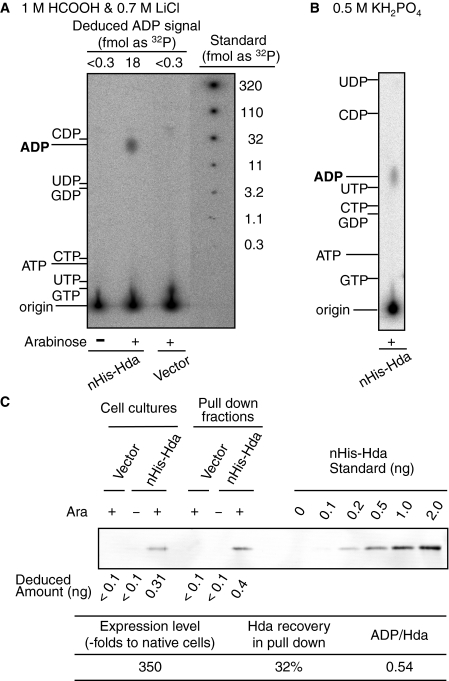
FIGURE 5.
Nucleotide-bound state of Hda in vivo. MC1061 cells harboring pBAD/HisB (Vector) or pBAD/nHis-Hda (nHis-Hda) were grown at 37 °C in a minimum medium containing [32P]orthophosphate. Aliquots (0.5 ml) were collected before (–) or after (+) incubation for 10 min in the presence of 0.5% arabinose, and used for a pulldown analysis (A and B) and Western bolt analysis (C). A and B, nHis-Hda was recovered from the cell lysates using a nickel pulldown method, resulting in the pulldown fractions (5 μl). Portions (0.25 μl) of the fractions were analyzed for Hda-bound nucleotides by TLC using a solvent consisting of 1 m HCOOH and 0.7 m LiCl (A) or 0.5 m KH2PO4 (B). The migration positions of various nucleotides were analyzed on the same TLC plate and detected by UV exposure and are indicated. Signals corresponding to ADP were quantified, and the amounts as a [32P]orthophosphate were deduced using a quantitative standard of [32P]orthophosphate spotted on the same TLC plate (Deduced ADP signal)(A). C, expression level and recovered amounts of nHis-Hda were determined by Western blot analysis. Labeled cells in the culture (5 μl) were used to determine the expression level, and pulldown fractions (0.2 μl) were isolated and used to determine the amount of recovered nHis-Hda. Intensities corresponding to nHis-Hda were quantified, and the amounts were deduced using a quantitative standard of purified nHis-Hda (Deduced amount). The deduced amount of nHis-Hda in the cells was used to calculate a relative value (Expression level) compared with the cellular amounts of Hda in MG1655 (25). The deduced amount of nHis-Hda in the pulldown fraction was used to calculate the recovery (Hda recovery in pull down), which is also shown. Based on the amount of [32P]ADP determined in A, the ratio of ADP molecules per nHis-Hda monomer in the pulldown fraction was determined (ADP/Hda).