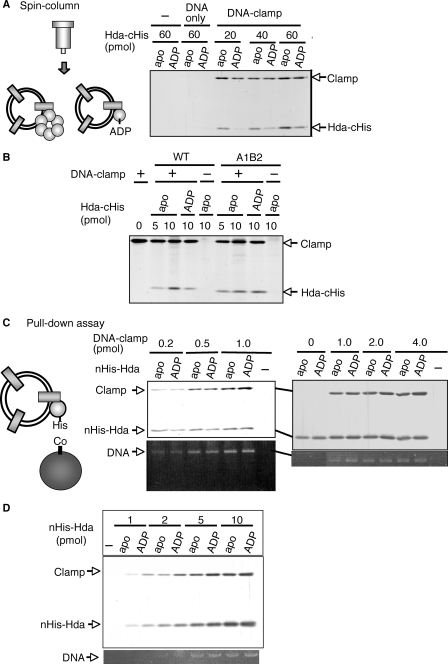
FIGURE 8.
Interaction of apo-Hda and ADP-Hda with the DNA-loaded clamp. A, spin-column analysis. Hda-cHis multimers (apo-Hda-cHis, apo) were preincubated at 30 °C for 20 min in the presence of 100 μm ADP, resulting in ADP-Hda-cHis (ADP). The indicated amounts of apo-Hda-cHis or ADP-Hda-cHis were incubated on ice for 15 min in the presence (DNA-clamp) or absence (–) of the DNA-loaded clamp (4 pmol as clamp, 320 ng as M13mp18 nicked circular DNA). Proteins associated with the DNA were isolated by gel filtration spin column into the void fraction. 1/10th of the void fraction was analyzed by SDS-PAGE and silver staining. The same experiment was performed using M13mp18 nicked circular DNA (320 ng) instead of the DNA-loaded clamp (DNA only). B, clamp binding activity of the Hda A1B2 mutant. Wild-type (WT) and A1B2 mutant (A1B2) Hda-cHis multimers were used as the apo-form (apo) or were preincubated in the presence of ADP as above (ADP). The indicated amounts of apo-Hda-cHis or ADP-preincubated Hda-cHis were incubated on ice for 15 min in the presence (+) or absence (–) of the DNA-loaded clamp (1 pmol as clamp). The void fraction was isolated by a gel filtration spin column and analyzed as described for A. C, pulldown assay. nHis-Hda multimers were used as the apo-form (apo) or were preincubated in the presence of ADP as above (ADP). TALON beads were incubated in the presence or absence (–) of 5 pmol of apo-nHis-Hda or ADP-nHis-Hda, followed by incubation on ice for 15 min in buffer containing the indicated amounts of the DNA-loaded clamp. The bead-bound materials were recovered, washed, and eluted in 1% SDS. An aliquot of the elution was analyzed by SDS-PAGE and silver staining (upper panel). The remaining elution was used for analysis of DNA recovery by 1% agarose gel electrophoresis and ethidium bromide staining (lower panel). D, various amounts of apo-nHis-Hda or ADP-nHis-Hda were analyzed by a similar pulldown assay in the presence of the DNA-loaded clamp (1 pmol).