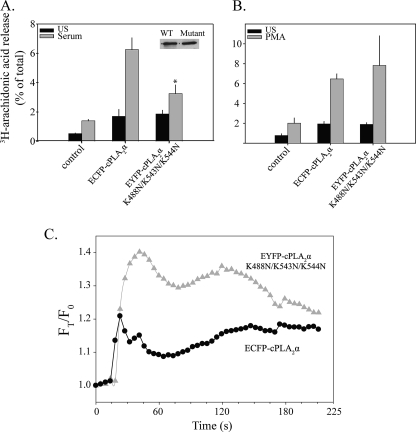
FIGURE 10.
Role of basic residues in cPLA2α catalytic domain in regulating translocation and AA release in response to serum and PMA. [3H]AA-labeled IMLF-/- expressing either wild type ECFP-cPLA2α or EYFP-cPLA2αK488N/K543N/K544N were stimulated for 10 min with serum (A) or 45 min with PMA (B), and [3H]AA release was measured and expressed as a percentage of the total cellular radioactivity in each well. The release of [3H]AA by the mutant was significantly less (p < 0.05) than by wild type cPLA2α, as indicated (*). Cell lysates were made from each well, and immunoblotting for cPLA2α was conducted to determine expression levels (inset) of wild type and mutant cPLA2α in each well. C, IMLF-/- co-expressing wild type ECFP-cPLA2α and EYFP-cPLA2αK488N/K543N/K544N were stimulated with serum, and images were collected using both a CFP and YFP filter and a ×40 oil immersion objective. Translocation data were calculated based on average fluorescence intensity of a mask of the Golgi in each cell. Values were calculated by subtracting background fluorescence and correcting for differential bleaching at each wavelength throughout the imaging. Data are presented relative to time 0 (FT/F0). Data are representative of 10 individual cells from three independent experiments.