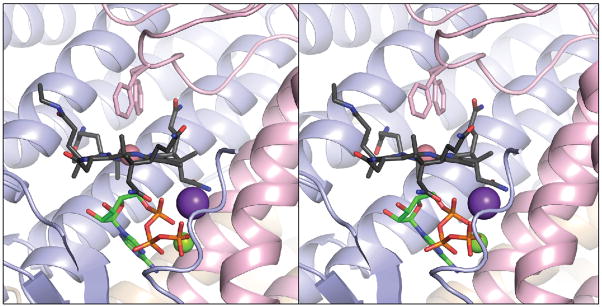
FIGURE 4.
The LrPduOΔ183 variant retains the ability to generate a four-coordinate intermediate. Stereoview of LrPduOΔ183 with the individual subunits of the trimer represented as ribbons colored in blue, red and brown. For clarity, only the active site at the interface of the red and blue subunits is shown. The carbon atoms of cob(II)alamin are colored in black while the carbon atoms of ATP are colored in green. The potassium (purple sphere) and magnesium (green sphere) atoms are also displayed. Residue Phe112 adopts two alternate conformations in this structure. Both conformations are displayed. The electron density corresponding to the DMB portion of cob(II)alamin is disordered and, therefore, DMB could not be modeled.