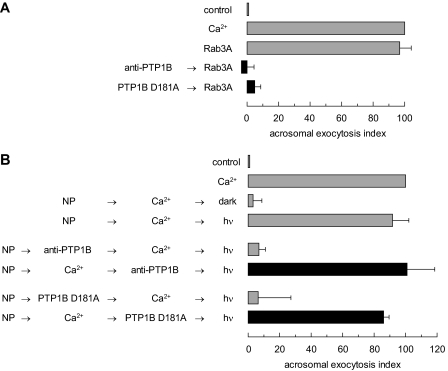
FIGURE 3.
PTP1B is required for human sperm AR after Rab3A but before intra-acrosomal calcium efflux. A, SLO-permeabilized human sperm were treated for 15 min at 37 °C in the presence of 3.3 nm anti-PTP1B antibodies or 300 nm PTP1B D181A. Acrosomal exocytosis was evaluated by FITC-PSA binding after an additional 15-min incubation at 37 °C in the absence or presence of 0.5 mm CaCl2 or 300 nm GTPγS-bound Rab3A (Rab3A). Controls (gray bars) included AR triggered by 300 nm GTPγS-bound Rab3A. B, SLO-permeabilized human sperm were loaded with 10 μm NP-EGTA-AM for 10 min at 37 °C to chelate intra-acrosomal calcium. AR was subsequently initiated by adding 0.5 mm CaCl2. After a further 10 min at 37 °C to allow exocytosis to proceed up to the intra-acrosomal calcium-sensitive step, sperm were treated for 10 min at 37 °C with anti-PTP1B antibodies or PTP1B D181A as in A. These procedures were carried out in the dark. UV photolysis of the chelator was induced at the end of the incubation period (NP → Ca2+ → anti-PTP1B/PTP1B D181A → hv, black bars). Sperm were fixed and AR was measured by FITC-PSA binding as described under “Experimental Procedures.” Several controls were run (gray bars): inhibitory effect of NP-EGTA-AM in the dark (NP → Ca2+ → dark); and the recovery upon illumination (NP → Ca2+ → hv); and the inhibitory effect of the antibodies and PTP1B D181A when present throughout the experiment (NP → anti-PTP1B/PTP1B D181A → Ca2+ → hv). The data were normalized as described under “Experimental Procedures.” Actual percentages of reacted sperm for control and Ca2+ ranged between 8–23 and 15–34%, respectively. The data represent the mean ± S.E. of at least three independent experiments.