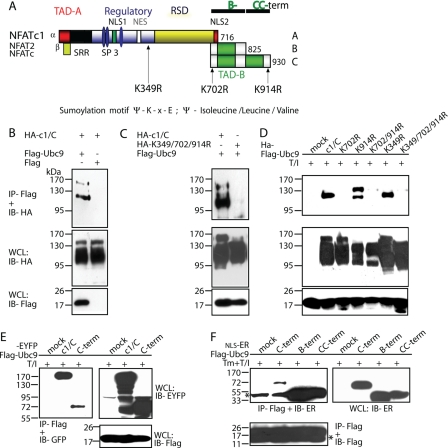
FIGURE 1.
NFATc1/C harbors three SUMO consensus motifs which facilitate interaction with Ubc9. A, schematic representation of NFATc1 isoforms and the SUMO consensus motifs (Ψ-Lys-X-Glu; Ψ= isoleucine/leucine/valine) shown by arrows. The short isoforms harbor only the common Lys-349 site, whereas NFATc1/C contains sites at Lys-702 and -914 in addition. Lysines to arginine exchanges create ΔSUMO mutations. The mutation of both C-terminal lysines is designated as K702R/K914R and of all three sites as K349R/K702R/K914R. An indication for the B-term and CC-term peptides are given. TAD, transactivation domain; RSD, Rel similarity domain; NLS, nuclear localization signal; NES, nuclear export signal; SRR and SP, Ser/Thr phosphorylation sites, respectively. B, NFATc1/C physically interacts with Ubc9 in vivo. HA-tagged c1/C-coding vector was transfected into 293T HEK cells, either in combination with pcDNA expressing FLAG-tagged Ubc9 or FLAG only. After 6 h of stimulation with T/I and a total of 48 h, protein lysates were prepared and subjected to anti-FLAG IP followed by immunoblot detection with anti-HA. Whole cell lysates (WCL) were also directly analyzed by immunoblotting with anti-HA and anti-FLAG, respectively. C, the SUMO site-deficient mutant K349R/K702R/K914R cannot recruit Ubc9. Procedures were the same as in B, except that K349R/K702R/K914R was included. D, the SUMO site at Lys-702 is the most relevant. Procedures were the same as in C but including all different SUMO site deficient mutants. E, the C terminus of NFATc1/C is sufficient for binding Ubc9. Procedures were the same as before, but EYFP-tagged NFATc1/C or only the C terminus were analyzed. F, both the NFATc1/B specific (B-term) and the NFATc1/C only (CC-term) C termini interact with Ubc9. Procedures were as before, but by use of constructs with nuclear localization signal-estrogen receptor fusions (estrogen receptor α), which are released for nuclear localization by addition of 4-hydroxytamoxifen. *, heavy or light chain of the precipitation antibody.