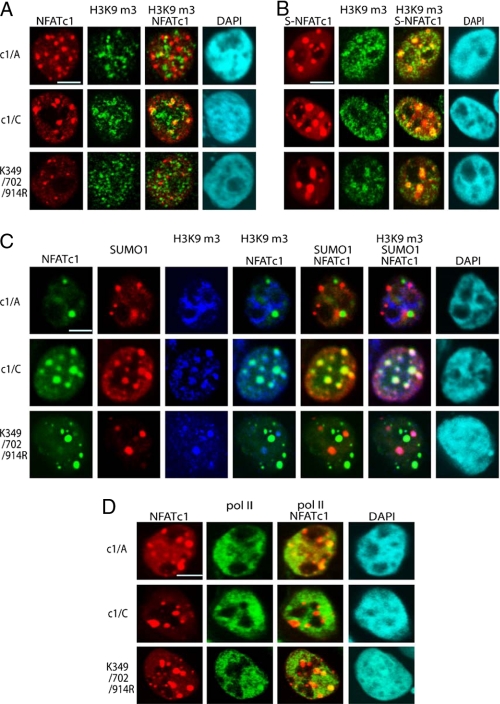
FIGURE 7.
Sumoylation directs NFATc1 into transcriptionally inactive chromatin. A, 293T HEK cells were transfected with plasmids encoding pHA-NFATc1/A, -c1/C, and -K349R/K702R/K914R and stimulated with T/I+CaCl2 for 1 h. IF was performed to detect NFATc1 with anti-HA and heterochromatin with anti-H3K9m3. Colocalization was analyzed by confocal microscopy. The scale bar represents 10 μm. B, 293T HEK cells were transfected with plasmids encoding SUMO1(S)-c1/A, -c1/C, and - K349R/K702R/K914R, which were C-terminally fused to EYFP. Cells were stimulated with T/I+CaCl2 for 1 h. IF was performed with anti-H3K9m3, and colocalization of SUMO-fused NFATc1 (S-NFATc1) and heterochromatin were analyzed by confocal microscopy. C, FLAG-SUMO1 was transfected along with c1/A, c1/C, and K349R/K702R/K914R plasmids, C-terminally fused to EYFP into 293T HEK cells, and stimulated with T/I+CaCl2 for 1 h followed by IF with anti-SUMO and anti-H3K9m3. Tri-colocalization of NFATc1, SUMO1, and H3K9m3 was analyzed by confocal microscopy. D, without sumoylation, NFATc1 colocalizes to hotspots of transcription. Transfection and stimulation was as in C. To reveal the colocalization pattern of NFATc1 and pol II-bodies, IF was performed with anti-pol II antibodies. The scale bar represents 10 μm. DAPI, 4′,6-diamidino-2-phenylindole.