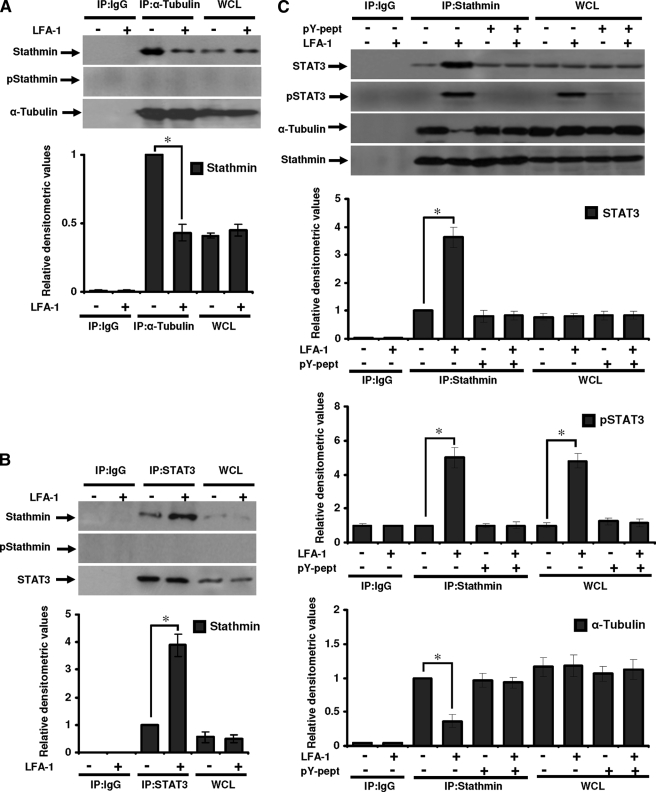
FIGURE 7.
STAT3 interaction with tubulin or stathmin in migrating T-cells. Serum-starved Hut78 cells were stimulated with or without anti-LFA-1 for 10 min and lysed. A, cell lysates (500 μg each) were immunoprecipitated with anti-α-tubulin (IP:α-Tubulin) or IgG (as isotype control; IP:IgG). Immunoprecipitates and whole cell lysates (used as control, 20 μg each; WCL) were Western blotted and probed with anti-stathmin, anti-phosphostathmin (Ser16; pStathmin), or anti-α-tubulin. B, cell lysates (500 μg each) were immunoprecipitated with anti-STAT3 (IP:STAT3) or IgG (IP:IgG). Immunoprecipates and whole cell lysates (20 μg each; WCL) were Western blotted and probed with anti-stathmin, anti-phospho-stathmin (Ser16; pStathmin), or anti-STAT3. C, untreated or pY-pept-treated Hut78 cells were stimulated with or without anti-LFA-1 for 10 min and lysed. Cell lysates (500 μg each) were immunoprecipitated with anti-stathmin (IP: Stathmin) or IgG (IP:IgG). Immunoprecipates and whole cell lysates (20 μg each; WCL) were Western blotted and probed with anti-STAT3, anti-phospho-STAT3 (Tyr705; pSTAT3), anti-α-tubulin, or anti-stathmin. Relative densitometric analysis of the individual band was performed and presented. Data are mean ± S.E. of three independent experiments. *, p < 0.05 with respect to corresponding controls.