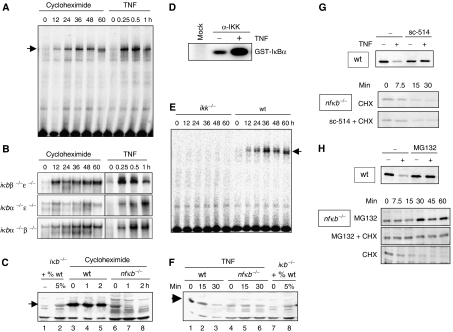
Figure 2.
Experimental studies of degradation pathways of NF-κB-bound and -free IκB proteins. (A) NF-κB activity as measured by EMSA of nuclear extracts from wild-type cells treated with 10 μg/ml CHX or 1 ng/ml TNF for indicated times. (B) NF-κB activity as measured by EMSA of nuclear extracts from ikbβ−/−ɛ−/−, ikbα−/−ɛ−/−, or ikbα−/−β−/− cells treated with 10 μg/ml CHX or 1 ng/ml TNF. (C) Western blot for IκBα in CHX-treated wild-type and nfkb−/− cells. The first two lanes show iκbα−/−β−/−ɛ−/− extract and iκbα−/−β−/−ɛ−/− extract mixed with 5% wild-type extract to show the protein level of IκBα in the nfkb−/− cells was approximately 5% that in the wild-type cells at time zero. (D) Cytoplasmic extracts of wild-type cells were immunoprecipitated with IKKγ antibody and subject to an in vitro kinase assay. In the ‘mock' lane, no antibody was added during the IP. (E) NF-κB activity as measured by EMSA of nuclear extracts from ikkα−/−β−/− or wild-type MEFs treated with 10 μg/ml CHX. (F) Western blot for IκBα of protein extracts from TNF (1 ng/ml)-treated wild-type or rela−/−crel−/−nfkb1−/− cells. (G) Western blots for IκBα of protein extracts from TNF-treated wild-type cells (top panel) in the presence or absence of the IKK-inhibitor sc-514. Bottom panels show Western blots for IκBα of protein extracts from CHX (10 μg/ml)-treated cells in the presence or absence of sc-514. (H) Western blots for IκBα of protein extracts from wild-type cells treated with TNFα (1 ng/ml) with or without the presence of the proteasome inhibitor MG132 (top panel). Western blots for IκBα of protein extracts from nfkb−/− cells treated with 10 μg/ml CHX, 10 μM MG132, or both.