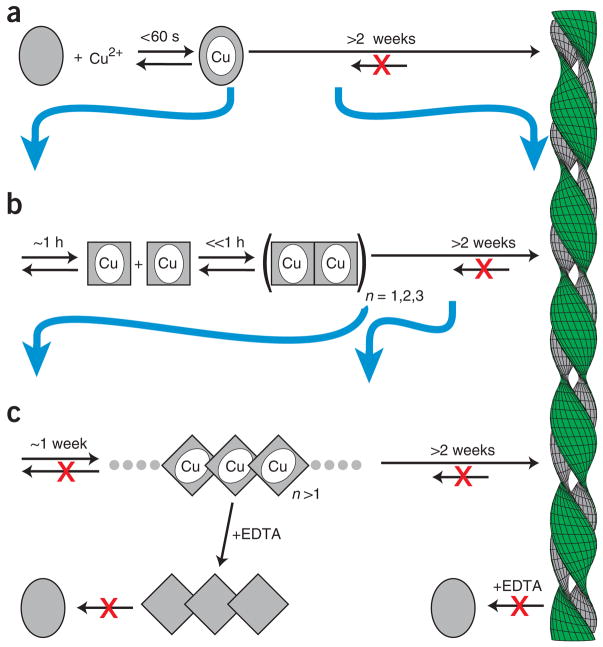
Figure 1.
Model of Cu2+-dependent amyloid formation of β2m. (a) In the absence of metal, β2m exists as a stable monomer (gray oval). The Cu2+ holo form leads to amyloid formation on a timescale of weeks15. (b) The initial Cu2+ binding event is followed by oligomerization on a timescale of ~1 h. The rate-limiting step of this oligomerization is a conformational rearrangement (gray rectangle)13. These oligomers require Cu2+ for stability and dissociate to monomeric form upon addition of a metal chelate (EDTA). (c) Cu2+ acts catalytically, giving rise to chelate-irreversible oligomers in a process that is accelerated by subdenaturing levels of urea comparable to that found in uremic patients28. Chelate resistance persists within the mature fiber.